Effexor
Rx only
Suicidality and Antidepressant Drugs
Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies of Major Depressive Disorder (MDD) and other psychiatric disorders. Anyone considering the use of Effexor XR or any other antidepressant in a child, adolescent, or young adult must balance this risk with the clinical need. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction in risk with antidepressants compared to placebo in adults aged 65 and older. Depression and certain other psychiatric disorders are themselves associated with increases in the risk of suicide. Patients of all ages who are started on antidepressant therapy should be monitored appropriately and observed closely for clinical worsening, suicidality, or unusual changes in behavior. Families and caregivers should be advised of the need for close observation and communication with the prescriber. Effexor XR is not approved for use in pediatric patients. (See WARNINGS: Clinical Worsening and Suicide Risk, PRECAUTIONS: Information for Patients, and PRECAUTIONS: Pediatric Use)
DESCRIPTION
Effexor XR is an extended-release capsule for oral administration that contains venlafaxine hydrochloride, a structurally novel antidepressant. It is designated (R/S)-1-[2-(dimethylamino)-1-(4-methoxyphenyl)ethyl] cyclohexanol hydrochloride or (±)-1-[α- [(dimethylamino)methyl]-p-methoxybenzyl] cyclohexanol hydrochloride and has the empirical formula of C17H27NO2 HCl. Its molecular weight is 313.87. The structural formula is shown below.
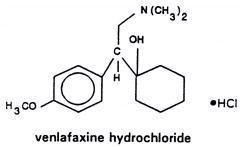
Venlafaxine hydrochloride is a white to off-white crystalline solid with a solubility of 572 mg/mL in water (adjusted to ionic strength of 0.2 M with sodium chloride). Its octanol:water (0.2 M sodium chloride) partition coefficient is 0.43.
Effexor XR is formulated as an extended-release capsule for once-a-day oral administration. Drug release is controlled by diffusion through the coating membrane on the spheroids and is not pH dependent. Capsules contain venlafaxine hydrochloride equivalent to 37.5 mg, 75 mg, or 150 mg venlafaxine. Inactive ingredients consist of cellulose, ethylcellulose, gelatin, hypromellose, iron oxide, and titanium dioxide.
CLINICAL PHARMACOLOGY
Pharmacodynamics
The mechanism of the antidepressant action of venlafaxine in humans is believed to be associated with its potentiation of neurotransmitter activity in the CNS. Preclinical studies have shown that venlafaxine and its active metabolite, O-desmethylvenlafaxine (ODV), are potent inhibitors of neuronal serotonin and norepinephrine reuptake and weak inhibitors of dopamine reuptake. Venlafaxine and ODV have no significant affinity for muscarinic cholinergic, H1-histaminergic, or α1-adrenergic receptors in vitro. Pharmacologic activity at these receptors is hypothesized to be associated with the various anticholinergic, sedative, and cardiovascular effects seen with other psychotropic drugs. Venlafaxine and ODV do not possess monoamine oxidase (MAO) inhibitory activity.
Pharmacokinetics
Steady-state concentrations of venlafaxine and ODV in plasma are attained within 3 days of oral multiple dose therapy. Venlafaxine and ODV exhibited linear kinetics over the dose range of 75 to 450 mg/day. Mean±SD steady-state plasma clearance of venlafaxine and ODV is 1.3±0.6 and 0.4±0.2 L/h/kg, respectively; apparent elimination half-life is 5±2 and 11±2 hours, respectively; and apparent (steady-state) volume of distribution is 7.5±3.7 and 5.7±1.8 L/kg, respectively. Venlafaxine and ODV are minimally bound at therapeutic concentrations to plasma proteins (27% and 30%, respectively).
Absorption
Venlafaxine is well absorbed and extensively metabolized in the liver. O-desmethylvenlafaxine (ODV) is the only major active metabolite. On the basis of mass balance studies, at least 92% of a single oral dose of venlafaxine is absorbed. The absolute bioavailability of venlafaxine is about 45%.
Administration of Effexor XR (150 mg q24 hours) generally resulted in lower Cmax (150 ng/mL for venlafaxine and 260 ng/mL for ODV) and later Tmax (5.5 hours for venlafaxine and 9 hours for ODV) than for Effexor (immediate release) [Cmax's for immediate release 75 mg q12 hours were 225 ng/mL for venlafaxine and 290 ng/mL for ODV; Tmax's were 2 hours for venlafaxine and 3 hours for ODV]. When equal daily doses of venlafaxine were administered as either an immediate release tablet or the extended-release capsule, the exposure to both venlafaxine and ODV was similar for the two treatments, and the fluctuation in plasma concentrations was slightly lower with the Effexor XR capsule. Effexor XR, therefore, provides a slower rate of absorption, but the same extent of absorption compared with the immediate release tablet.
Food did not affect the bioavailability of venlafaxine or its active metabolite, ODV. Time of administration (AM vs PM) did not affect the pharmacokinetics of venlafaxine and ODV from the 75 mg Effexor XR capsule.
Metabolism and Excretion
Following absorption, venlafaxine undergoes extensive presystemic metabolism in the liver, primarily to ODV, but also to N-desmethylvenlafaxine, N,O-didesmethylvenlafaxine, and other minor metabolites. In vitro studies indicate that the formation of ODV is catalyzed by CYP2D6; this has been confirmed in a clinical study showing that patients with low CYP2D6 levels (“poor metabolizers”) had increased levels of venlafaxine and reduced levels of ODV compared to people with normal CYP2D6 (“extensive metabolizers”). The differences between the CYP2D6 poor and extensive metabolizers, however, are not expected to be clinically important because the sum of venlafaxine and ODV is similar in the two groups and venlafaxine and ODV are pharmacologically approximately equiactive and equipotent.
Approximately 87% of a venlafaxine dose is recovered in the urine within 48 hours as unchanged venlafaxine (5%), unconjugated ODV (29%), conjugated ODV (26%), or other minor inactive metabolites (27%). Renal elimination of venlafaxine and its metabolites is thus the primary route of excretion.
Special Populations
Age and Gender: A population pharmacokinetic analysis of 404 venlafaxine-treated patients from two studies involving both b.i.d. and t.i.d. regimens showed that dose-normalized trough plasma levels of either venlafaxine or ODV were unaltered by age or gender differences. Dosage adjustment based on the age or gender of a patient is generally not necessary (see DOSAGE AND ADMINISTRATION).
Extensive/Poor Metabolizers: Plasma concentrations of venlafaxine were higher in CYP2D6 poor metabolizers than extensive metabolizers. Because the total exposure (AUC) of venlafaxine and ODV was similar in poor and extensive metabolizer groups, however, there is no need for different venlafaxine dosing regimens for these two groups.
Liver Disease: In 9 subjects with hepatic cirrhosis, the pharmacokinetic disposition of both venlafaxine and ODV was significantly altered after oral administration of venlafaxine. Venlafaxine elimination half-life was prolonged by about 30%, and clearance decreased by about 50% in cirrhotic subjects compared to normal subjects. ODV elimination half-life was prolonged by about 60%, and clearance decreased by about 30% in cirrhotic subjects compared to normal subjects. A large degree of intersubject variability was noted. Three patients with more severe cirrhosis had a more substantial decrease in venlafaxine clearance (about 90%) compared to normal subjects.
In a second study, venlafaxine was administered orally and intravenously in normal (n = 21) subjects, and in Child-Pugh A (n = 8) and Child-Pugh B (n = 11) subjects (mildly and moderately impaired, respectively). Venlafaxine oral bioavailability was increased 2-3 fold, oral elimination half-life was approximately twice as long and oral clearance was reduced by more than half, compared to normal subjects. In hepatically impaired subjects, ODV oral elimination half-life was prolonged by about 40%, while oral clearance for ODV was similar to that for normal subjects. A large degree of intersubject variability was noted.
Dosage adjustment is necessary in these hepatically impaired patients (see DOSAGE AND ADMINISTRATION).
Renal Disease: In a renal impairment study, venlafaxine elimination half-life after oral administration was prolonged by about 50% and clearance was reduced by about 24% in renally impaired patients (GFR=10 to 70 mL/min), compared to normal subjects. In dialysis patients, venlafaxine elimination half-life was prolonged by about 180% and clearance was reduced by about 57% compared to normal subjects. Similarly, ODV elimination half-life was prolonged by about 40% although clearance was unchanged in patients with renal impairment (GFR=10 to 70 mL/min) compared to normal subjects. In dialysis patients, ODV elimination half-life was prolonged by about 142% and clearance was reduced by about 56% compared to normal subjects. A large degree of intersubject variability was noted. Dosage adjustment is necessary in these patients (see DOSAGE AND ADMINISTRATION).
Clinical Trials
Major Depressive Disorder
The efficacy of Effexor XR (venlafaxine hydrochloride) extended-release capsules as a treatment for major depressive disorder was established in two placebo-controlled, short-term, flexible-dose studies in adult outpatients meeting DSM-III-R or DSM-IV criteria for major depressive disorder.
A 12-week study utilizing Effexor XR doses in a range 75 to 150 mg/day (mean dose for completers was 136 mg/day) and an 8-week study utilizing Effexor XR doses in a range 75 to 225 mg/day (mean dose for completers was 177 mg/day) both demonstrated superiority of Effexor XR over placebo on the HAM-D total score, HAM-D Depressed Mood Item, the MADRS total score, the Clinical Global Impressions (CGI) Severity of Illness item, and the CGI Global Improvement item. In both studies, Effexor XR was also significantly better than placebo for certain factors of the HAM-D, including the anxiety/somatization factor, the cognitive disturbance factor, and the retardation factor, as well as for the psychic anxiety score.
A 4-week study of inpatients meeting DSM-III-R criteria for major depressive disorder with melancholia utilizing Effexor (immediate release) in a range of 150 to 375 mg/day (t.i.d. schedule) demonstrated superiority of Effexor over placebo. The mean dose in completers was 350 mg/day.
Examination of gender subsets of the population studied did not reveal any differential responsiveness on the basis of gender.
In one longer-term study, adult outpatients meeting DSM-IV criteria for major depressive disorder who had responded during an 8-week open trial on Effexor XR (75, 150, or 225 mg, qAM) were randomized to continuation of their same Effexor XR dose or to placebo, for up to 26 weeks of observation for relapse. Response during the open phase was defined as a CGI Severity of Illness item score of ≤3 and a HAM-D-21 total score of ≤10 at the day 56 evaluation. Relapse during the double-blind phase was defined as follows: (1) a reappearance of major depressive disorder as defined by DSM-IV criteria and a CGI Severity of Illness item score of ≥4 (moderately ill), (2) 2 consecutive CGI Severity of Illness item scores of ≥4, or (3) a final CGI Severity of Illness item score of ≥4 for any patient who withdrew from the study for any reason. Patients receiving continued Effexor XR treatment experienced significantly lower relapse rates over the subsequent 26 weeks compared with those receiving placebo.
In a second longer-term trial, adult outpatients meeting DSM-III-R criteria for major depressive disorder, recurrent type, who had responded (HAM-D-21 total score ≤12 at the day 56 evaluation) and continued to be improved [defined as the following criteria being met for days 56 through 180: (1) no HAM-D-21 total score ≥20; (2) no more than 2 HAM-D-21 total scores >10, and (3) no single CGI Severity of Illness item score ≥4 (moderately ill)] during an initial 26 weeks of treatment on Effexor (immediate release) [100 to 200 mg/day, on a b.i.d. schedule] were randomized to continuation of their same Effexor dose or to placebo. The follow-up period to observe patients for relapse, defined as a CGI Severity of Illness item score ≥4, was for up to 52 weeks. Patients receiving continued Effexor treatment experienced significantly lower relapse rates over the subsequent 52 weeks compared with those receiving placebo.
Generalized Anxiety Disorder
The efficacy of Effexor XR capsules as a treatment for Generalized Anxiety Disorder (GAD) was established in two 8-week, placebo-controlled, fixed-dose studies, one 6-month, placebo-controlled, fixed-dose study, and one 6-month, placebo-controlled, flexible-dose study in adult outpatients meeting DSM-IV criteria for GAD.
One 8-week study evaluating Effexor XR doses of 75, 150, and 225 mg/day, and placebo showed that the 225 mg/day dose was more effective than placebo on the Hamilton Rating Scale for Anxiety (HAM-A) total score, both the HAM-A anxiety and tension items, and the Clinical Global Impressions (CGI) scale. While there was also evidence for superiority over placebo for the 75 and 150 mg/day doses, these doses were not as consistently effective as the highest dose. A second 8-week study evaluating Effexor XR doses of 75 and 150 mg/day and placebo showed that both doses were more effective than placebo on some of these same outcomes; however, the 75 mg/day dose was more consistently effective than the 150 mg/day dose. A dose-response relationship for effectiveness in GAD was not clearly established in the 75 to 225 mg/day dose range utilized in these two studies.
Two 6-month studies, one evaluating Effexor XR doses of 37.5, 75, and 150 mg/day and the other evaluating Effexor XR doses of 75 to 225 mg/day, showed that daily doses of 75 mg or higher were more effective than placebo on the HAM-A total, both the HAM-A anxiety and tension items, and the CGI scale during 6 months of treatment. While there was also evidence for superiority over placebo for the 37.5 mg/day dose, this dose was not as consistently effective as the higher doses.
Examination of gender subsets of the population studied did not reveal any differential responsiveness on the basis of gender.
Social Anxiety Disorder (Social Phobia)
The efficacy of Effexor XR capsules as a treatment for Social Anxiety Disorder (also known as Social Phobia) was established in four double-blind, parallel-group, 12-week, multicenter, placebo-controlled, flexible-dose studies and one double-blind, parallel-group, 6-month, placebo-controlled, fixed/flexible-dose study in adult outpatients meeting DSM-IV criteria for Social Anxiety Disorder. Patients received doses in a range of 75 to 225 mg/day. Efficacy was assessed with the Liebowitz Social Anxiety Scale (LSAS). In these five trials, Effexor XR was significantly more effective than placebo on change from baseline to endpoint on the LSAS total score. There was no evidence for any greater effectiveness of the 150 to 225 mg/day group compared to the 75 mg/day group in the 6-month study.
Examination of subsets of the population studied did not reveal any differential responsiveness on the basis of gender. There was insufficient information to determine the effect of age or race on outcome in these studies.
Panic Disorder
The efficacy of Effexor XR capsules as a treatment for panic disorder was established in two double-blind, 12-week, multicenter, placebo-controlled studies in adult outpatients meeting DSM-IV criteria for panic disorder, with or without agoraphobia. Patients received fixed doses of 75 or 150 mg/day in one study and 75 or 225 mg/day in the other study.
Efficacy was assessed on the basis of outcomes in three variables: (1) percentage of patients free of full-symptom panic attacks on the Panic and Anticipatory Anxiety Scale (PAAS); (2) mean change from baseline to endpoint on the Panic Disorder Severity Scale (PDSS) total score; and (3) percentage of patients rated as responders (much improved or very much improved) on the Clinical Global Impressions (CGI) Improvement scale. In these two trials, Effexor XR was significantly more effective than placebo in all three variables.
In the two 12-week studies described above, one evaluating Effexor XR doses of 75 and 150 mg/day and the other evaluating Effexor XR doses of 75 and 225 mg/day, efficacy was established for each dose. A dose-response relationship for effectiveness in patients with panic disorder was not clearly established in fixed-dose studies.
Examination of subsets of the population studied did not reveal any differential responsiveness on the basis of gender. There was insufficient information to determine the effect of age or race on outcome in these studies.
In a longer-term study, adult outpatients meeting DSM-IV criteria for panic disorder who had responded during a 12-week open phase with Effexor XR (75 to 225 mg/day) were randomly assigned to continue the same Effexor XR dose (75, 150, or 225 mg) or switch to placebo for observation for relapse under double-blind conditions. Response during the open phase was defined as ≤ 1 full-symptom panic attack per week during the last 2 weeks of the open phase and a CGI Improvement score of 1 (very much improved) or 2 (much improved). Relapse during the double-blind phase was defined as having 2 or more full-symptom panic attacks per week for 2 consecutive weeks or having discontinued due to loss of effectiveness as determined by the investigators during the study. Randomized patients were in response status for a mean time of 34 days prior to being randomized. In the randomized phase following the 12-week open-label period, patients receiving continued Effexor XR experienced a significantly longer time to relapse.
INDICATIONS AND USAGE
Major Depressive Disorder
Effexor XR (venlafaxine hydrochloride) extended-release capsules is indicated for the treatment of major depressive disorder.
The efficacy of Effexor XR in the treatment of major depressive disorder was established in 8- and 12-week controlled trials of adult outpatients whose diagnoses corresponded most closely to the DSM-III-R or DSM-IV category of major depressive disorder (see Clinical Trials).
A major depressive episode (DSM-IV) implies a prominent and relatively persistent (nearly every day for at least 2 weeks) depressed mood or the loss of interest or pleasure in nearly all activities, representing a change from previous functioning, and includes the presence of at least five of the following nine symptoms during the same two-week period: depressed mood, markedly diminished interest or pleasure in usual activities, significant change in weight and/or appetite, insomnia or hypersomnia, psychomotor agitation or retardation, increased fatigue, feelings of guilt or worthlessness, slowed thinking or impaired concentration, a suicide attempt or suicidal ideation.
The efficacy of Effexor (immediate release) in the treatment of major depressive disorder in adult inpatients meeting diagnostic criteria for major depressive disorder with melancholia was established in a 4-week controlled trial (see Clinical Trials). The safety and efficacy of Effexor XR in hospitalized depressed patients have not been adequately studied.
The efficacy of Effexor XR in maintaining a response in major depressive disorder for up to 26 weeks following 8 weeks of acute treatment was demonstrated in a placebo-controlled trial. The efficacy of Effexor (immediate release) in maintaining a response in patients with recurrent major depressive disorder who had responded and continued to be improved during an initial 26 weeks of treatment and were then followed for a period of up to 52 weeks was demonstrated in a second placebo-controlled trial (see Clinical Trials). Nevertheless, the physician who elects to use Effexor/Effexor XR for extended periods should periodically re-evaluate the long-term usefulness of the drug for the individual patient (see DOSAGE AND ADMINISTRATION).
Generalized Anxiety Disorder
Effexor XR is indicated for the treatment of Generalized Anxiety Disorder (GAD) as defined in DSM-IV. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic.
The efficacy of Effexor XR in the treatment of GAD was established in 8-week and 6-month placebo-controlled trials in adult outpatients diagnosed with GAD according to DSM-IV criteria (see Clinical Trials).
Generalized Anxiety Disorder (DSM-IV) is characterized by excessive anxiety and worry (apprehensive expectation) that is persistent for at least 6 months and which the person finds difficult to control. It must be associated with at least 3 of the following 6 symptoms: restlessness or feeling keyed up or on edge, being easily fatigued, difficulty concentrating or mind going blank, irritability, muscle tension, sleep disturbance.
Although the effectiveness of Effexor XR has been demonstrated in 6-month clinical trials in patients with GAD, the physician who elects to use Effexor XR for extended periods should periodically re-evaluate the long-term usefulness of the drug for the individual patient (see DOSAGE AND ADMINISTRATION).
Social Anxiety Disorder
Effexor XR is indicated for the treatment of Social Anxiety Disorder, also known as Social Phobia, as defined in DSM-IV (300.23).
Social Anxiety Disorder (DSM-IV) is characterized by a marked and persistent fear of 1 or more social or performance situations in which the person is exposed to unfamiliar people or to possible scrutiny by others. Exposure to the feared situation almost invariably provokes anxiety, which may approach the intensity of a panic attack. The feared situations are avoided or endured with intense anxiety or distress. The avoidance, anxious anticipation, or distress in the feared situation(s) interferes significantly with the person's normal routine, occupational or academic functioning, or social activities or relationships, or there is a marked distress about having the phobias. Lesser degrees of performance anxiety or shyness generally do not require psychopharmacological treatment.
The efficacy of Effexor XR in the treatment of Social Anxiety Disorder was established in four 12-week and one 6-month placebo-controlled trials in adult outpatients with Social Anxiety Disorder (DSM-IV) (see Clinical Trials).
Although the effectiveness of Effexor XR has been demonstrated in a 6-month clinical trial in patients with Social Anxiety Disorder, the physician who elects to use Effexor XR for extended periods should periodically re-evaluate the long-term usefulness of the drug for the individual patient (see DOSAGE AND ADMINISTRATION).
Panic Disorder
Effexor XR is indicated for the treatment of panic disorder, with or without agoraphobia, as defined in DSM-IV. Panic disorder is characterized by the occurrence of unexpected panic attacks and associated concern about having additional attacks, worry about the implications or consequences of the attacks, and/or a significant change in behavior related to the attacks.
Panic disorder (DSM-IV) is characterized by recurrent, unexpected panic attacks, ie, a discrete period of intense fear or discomfort, in which four (or more) of the following symptoms develop abruptly and reach a peak within 10 minutes: 1) palpitations, pounding heart, or accelerated heart rate; 2) sweating; 3) trembling or shaking; 4) sensations of shortness of breath or smothering; 5) feeling of choking; 6) chest pain or discomfort; 7) nausea or abdominal distress; 8) feeling dizzy, unsteady, lightheaded, or faint; 9) derealization (feelings of unreality) or depersonalization (being detached from oneself); 10) fear of losing control; 11) fear of dying; 12) paresthesias (numbness or tingling sensations); 13) chills or hot flushes.
The efficacy of Effexor XR in the treatment of panic disorder was established in two 12-week placebo-controlled trials in adult outpatients with panic disorder (DSM-IV). The efficacy of Effexor XR in prolonging time to relapse in panic disorder among responders following 12 weeks of open-label acute treatment was demonstrated in a placebo-controlled study (see CLINICAL PHARMACOLOGY, Clinical Trials). Nevertheless, the physician who elects to use Effexor XR for extended periods should periodically re-evaluate the long-term usefulness of the drug for the individual patient (see DOSAGE AND ADMINISTRATION).
CONTRAINDICATIONS
Hypersensitivity to venlafaxine hydrochloride or to any excipients in the formulation.
Concomitant use in patients taking monoamine oxidase inhibitors (MAOIs) is contraindicated (see WARNINGS).
WARNINGS
Clinical Worsening and Suicide Risk
Patients with major depressive disorder (MDD), both adult and pediatric, may experience worsening of their depression and/or the emergence of suicidal ideation and behavior (suicidality) or unusual changes in behavior, whether or not they are taking antidepressant medications, and this risk may persist until significant remission occurs. Suicide is a known risk of depression and certain other psychiatric disorders, and these disorders themselves are the strongest predictors of suicide. There has been a long standing concern, however, that antidepressants may have a role in inducing worsening of depression and the emergence of suicidality in certain patients during the early phases of treatment. Pooled analyses of short-term placebo-controlled trials of antidepressant drugs (SSRIs and others) showed that these drugs increase the risk of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults (ages 18-24) with major depressive disorder (MDD) and other psychiatric disorders. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction with antidepressants compared to placebo in adults aged 65 and older.
The pooled analyses of placebo-controlled trials in children and adolescents with MDD, obsessive compulsive disorder (OCD), or other psychiatric disorders included a total of 24 short-term trials of 9 antidepressant drugs in over 4400 patients. The pooled analyses of placebo-controlled trials in adults with MDD or other psychiatric disorders included a total of 295 short-term trials (median duration of 2 months) of 11 antidepressant drugs in over 77,000 patients. There was considerable variation in risk of suicidality among drugs, but a tendency toward an increase in the younger patients for almost all drugs studied. There were differences in absolute risk of suicidality across the different indications, with the highest incidence in MDD. The risk differences (drug vs placebo), however, were relatively stable within age strata and across indications. These risk differences (drug-placebo difference in the number of cases of suicidality per 1000 patients treated) are provided in Table 1.
| Age Range | Drug-Placebo Difference in Number of Cases of Suicidality per 1000 Patients Treated |
| Increases Compared to Placebo | |
| <18 | 14 additional cases |
| 18-24 | 5 additional cases |
| Decreases Compared to Placebo | |
| 25-64 | 1 fewer case |
| ≥65 | 6 fewer cases |
No suicides occurred in any of the pediatric trials. There were suicides in the adult trials, but the number was not sufficient to reach any conclusion about drug effect on suicide.
It is unknown whether the suicidality risk extends to longer-term use, i.e., beyond several months. However, there is substantial evidence from placebo-controlled maintenance trials in adults with depression that the use of antidepressants can delay the recurrence of depression.
All patients being treated with antidepressants for any indication should be monitored appropriately and observed closely for clinical worsening, suicidality, and unusual changes in behavior, especially during the initial few months of a course of drug therapy, or at times of dose changes, either increases or decreases.
The following symptoms, anxiety, agitation, panic attacks, insomnia, irritability, hostility, aggressiveness, impulsivity, akathisia (psychomotor restlessness), hypomania, and mania, have been reported in adult and pediatric patients being treated with antidepressants for major depressive disorder as well as for other indications, both psychiatric and nonpsychiatric. Although a causal link between the emergence of such symptoms and either the worsening of depression and/or the emergence of suicidal impulses has not been established, there is concern that such symptoms may represent precursors to emerging suicidality.
Consideration should be given to changing the therapeutic regimen, including possibly discontinuing the medication, in patients whose depression is persistently worse, or who are experiencing emergent suicidality or symptoms that might be precursors to worsening depression or suicidality, especially if these symptoms are severe, abrupt in onset, or were not part of the patient's presenting symptoms.
If the decision has been made to discontinue treatment, medication should be tapered, as rapidly as is feasible, but with recognition that abrupt discontinuation can be associated with certain symptoms (see PRECAUTIONS and DOSAGE AND ADMINISTRATION, Discontinuation of Treatment with Effexor XR, for a description of the risks of discontinuation of Effexor XR).
Families and caregivers of patients being treated with antidepressants for major depressive disorder or other indications, both psychiatric and nonpsychiatric, should be alerted about the need to monitor patients for the emergence of agitation, irritability, unusual changes in behavior, and the other symptoms described above, as well as the emergence of suicidality, and to report such symptoms immediately to health care providers. Such monitoring should include daily observation by families and caregivers. Prescriptions for Effexor XR should be written for the smallest quantity of capsules consistent with good patient management, in order to reduce the risk of overdose.
Screening Patients for Bipolar Disorder
A major depressive episode may be the initial presentation of bipolar disorder. It is generally believed (though not established in controlled trials) that treating such an episode with an antidepressant alone may increase the likelihood of precipitation of a mixed/manic episode in patients at risk for bipolar disorder. Whether any of the symptoms described above represent such a conversion is unknown. However, prior to initiating treatment with an antidepressant, patients with depressive symptoms should be adequately screened to determine if they are at risk for bipolar disorder; such screening should include a detailed psychiatric history, including a family history of suicide, bipolar disorder, and depression. It should be noted that Effexor XR is not approved for use in treating bipolar depression.
Potential for Interaction with Monoamine Oxidase Inhibitors
Adverse reactions, some of which were serious, have been reported in patients who have recently been discontinued from a monoamine oxidase inhibitor (MAOI) and started on venlafaxine, or who have recently had venlafaxine therapy discontinued prior to initiation of an MAOI. These reactions have included tremor, myoclonus, diaphoresis, nausea, vomiting, flushing, dizziness, hyperthermia with features resembling neuroleptic malignant syndrome, seizures, and death. In patients receiving antidepressants with pharmacological properties similar to venlafaxine in combination with an MAOI, there have also been reports of serious, sometimes fatal, reactions. For a selective serotonin reuptake inhibitor, these reactions have included hyperthermia, rigidity, myoclonus, autonomic instability with possible rapid fluctuations of vital signs, and mental status changes that include extreme agitation progressing to delirium and coma. Some cases presented with features resembling neuroleptic malignant syndrome. Severe hyperthermia and seizures, sometimes fatal, have been reported in association with the combined use of tricyclic antidepressants and MAOIs. These reactions have also been reported in patients who have recently discontinued these drugs and have been started on an MAOI. The effects of combined use of venlafaxine and MAOIs have not been evaluated in humans or animals. Therefore, because venlafaxine is an inhibitor of both norepinephrine and serotonin reuptake, it is recommended that Effexor XR (venlafaxine hydrochloride) extended-release capsules not be used in combination with an MAOI, or within at least 14 days of discontinuing treatment with an MAOI. Based on the half-life of venlafaxine, at least 7 days should be allowed after stopping venlafaxine before starting an MAOI.
Serotonin Syndrome or Neuroleptic Malignant Syndrome (NMS)-like Reactions
The development of a potentially life-threatening serotonin syndrome or Neuroleptic Malignant Syndrome (NMS)-like reactions have been reported with SNRIs and SSRIs alone, including Effexor XR treatment, but particularly with concomitant use of serotonergic drugs (including triptans) with drugs which impair metabolism of serotonin (including MAOIs), or with antipsychotics or other dopamine antagonists. Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, coma), autonomic instability (e.g., tachycardia, labile blood pressure, hyperthermia), neuromuscular aberrations (e.g., hyperreflexia, incoordination) and/or gastrointestinal symptoms [e.g., nausea, vomiting, diarrhea] (see PRECAUTIONS, Drug Interactions). Serotonin syndrome, in its most severe form can resemble neuroleptic malignant syndrome, which includes hyperthermia, muscle rigidity, autonomic instability with possible rapid fluctuation of vital signs, and mental status changes. Patients should be monitored for the emergence of serotonin syndrome or NMS-like signs and symptoms.
The concomitant use of Effexor XR with MAOIs intended to treat depression is contraindicated (see CONTRAINDICATIONS and WARNINGS, Potential for Interaction with Monoamine Oxidase Inhibitors).
If concomitant treatment of Effexor XR with a 5-hydroxytryptamine receptor agonist (triptan) is clinically warranted, careful observation of the patient is advised, particularly during treatment initiation and dose increases (see PRECAUTIONS, Drug Interactions).
The concomitant use of Effexor XR with serotonin precursors (such as tryptophan) is not recommended (see PRECAUTIONS, Drug Interactions).
Treatment with Effexor XR and any concomitant serotonergic or antidopaminergic agents, including antipsychotics, should be discontinued immediately if the above events occur and supportive symptomatic treatment should be initiated.
Sustained Hypertension
Effexor XR treatment is associated with sustained hypertension (defined as treatment-emergent supine diastolic blood pressure (SDBP) ≥90 mm Hg and ≥10 mm Hg above baseline for 3 consecutive on-therapy visits (see Table 2).
An analysis for patients in Effexor (immediate release) studies meeting criteria for sustained hypertension revealed a dose-dependent increase in the incidence of sustained hypertension for Effexor (immediate release) (see Table 3).
An insufficient number of patients received mean doses of Effexor XR over 300 mg/day to fully evaluate the incidence of sustained increases in blood pressure at these higher doses.
| MDD = major depressive disorder | |||
| MDD (75-375 mg/day) | GAD (37.5-225 mg/day) | Social Anxiety Disorder (75-225 mg/day) | Panic Disorder (75-225 mg/day) |
| 19/705 (3) | 5/1011 (0.5) | 5/771 (0.6) | 9/973 (0.9) |
| Effexor mg/day | Incidence |
| <100 | 3% |
| >100 to ≤200 | 5% |
| >200 to ≤300 | 7% |
| >300 | 13% |
In premarketing major depressive disorder studies, 0.7% (5/705) of the Effexor XR-treated patients discontinued treatment because of elevated blood pressure. Among these patients, most of the blood pressure increases were in a modest range (12 to 16 mm Hg, SDBP). In premarketing GAD studies up to 8 weeks and up to 6 months, 0.7% (10/1381) and 1.3% (7/535) of the Effexor XR-treated patients, respectively, discontinued treatment because of elevated blood pressure. Among these patients, most of the blood pressure increases were in a modest range (12 to 25 mm Hg, SDBP up to 8 weeks; 8 to 28 mm Hg up to 6 months). In premarketing Social Anxiety Disorder studies up to 6 months, 0.6% (5/771) of the Effexor XR-treated patients discontinued treatment because of elevated blood pressure. In these patients, the blood pressure increases were modest (1-24 mm Hg, SDBP). In premarketing panic disorder studies up to 12 weeks, 0.5% (5/1001) of the Effexor XR-treated patients discontinued treatment because of elevated blood pressure. In these patients, the blood pressure increases were in a modest range (7 to 19 mm Hg, SDBP).
Sustained increases of SDBP could have adverse consequences. Cases of elevated blood pressure requiring immediate treatment have been reported in post marketing experience. Pre-existing hypertension should be controlled before treatment with venlafaxine. It is recommended that patients receiving Effexor XR have regular monitoring of blood pressure. For patients who experience a sustained increase in blood pressure while receiving venlafaxine, either dose reduction or discontinuation should be considered.
Elevations in Systolic and Diastolic Blood Pressure
In placebo-controlled premarketing studies, there were changes in mean blood pressure (see Table 4 for mean changes in supine systolic and supine diastolic blood pressure). Across most indications, a dose-related increase in supine systolic and diastolic blood pressure was evident in Effexor XR-treated patients.
| 1 Supine Systolic Blood Pressure | ||||||
| Effexor XR mg/day | Placebo | |||||
| ≤75 | >75 | |||||
| SSBP1 | SDBP2 | SSBP | SDBP | SSBP | SDBP | |
| Major Depressive Disorder | ||||||
| 8-12 weeks | -0.28 | 0.37 | 2.93 | 3.56 | -1.08 | -0.10 |
| Generalized Anxiety Disorder | ||||||
| 8 weeks | -0.28 | 0.02 | 2.40 | 1.68 | -1.26 | -0.92 |
| 6 months | 1.27 | -0.69 | 2.06 | 1.28 | -1.29 | -0.74 |
| Social Anxiety Disorder | ||||||
| 12 weeks | -0.29 | -1.26 | 1.18 | 1.34 | -1.96 | -1.22 |
| 6 months | -0.98 | -0.49 | 2.51 | 1.96 | -1.84 | -0.65 |
| Panic Disorder | ||||||
| 10-12 weeks | -1.15 | 0.97 | -0.36 | 0.16 | -1.29 | -0.99 |
Across all clinical trials in MDD, GAD, Social Anxiety Disorder and panic disorder, 1.4% of patients in the Effexor XR-treated groups experienced a ≥15 mm Hg increase in supine diastolic blood pressure with blood pressure ≥105 mm Hg compared to 0.9% of patients in the placebo groups. Similarly, 1% of patients in the Effexor XR-treated groups experienced a ≥20 mm Hg increase in supine systolic blood pressure with blood pressure ≥180 mm Hg compared to 0.3% of patients in the placebo groups.
Mydriasis
Mydriasis has been reported in association with venlafaxine; therefore patients with raised intraocular pressure or those at risk of acute narrow-angle glaucoma (angle-closure glaucoma) should be monitored (see PRECAUTIONS, Information for Patients).
PRECAUTIONS
General
Discontinuation of Treatment with Effexor XR
Discontinuation symptoms have been systematically evaluated in patients taking venlafaxine, to include prospective analyses of clinical trials in Generalized Anxiety Disorder and retrospective surveys of trials in major depressive disorder, and Social Anxiety Disorder. Abrupt discontinuation or dose reduction of venlafaxine at various doses has been found to be associated with the appearance of new symptoms, the frequency of which increased with increased dose level and with longer duration of treatment. Reported symptoms include agitation, anorexia, anxiety, confusion, impaired coordination and balance, diarrhea, dizziness, dry mouth, dysphoric mood, fasciculation, fatigue, flu-like symptoms, headaches, hypomania, insomnia, nausea, nervousness, nightmares, sensory disturbances (including shock-like electrical sensations), somnolence, sweating, tremor, vertigo, and vomiting.
During marketing of Effexor XR, other SNRIs (Serotonin and Norepinephrine Reuptake Inhibitors), and SSRIs (Selective Serotonin Reuptake Inhibitors), there have been spontaneous reports of adverse events occurring upon discontinuation of these drugs, particularly when abrupt, including the following: dysphoric mood, irritability, agitation, dizziness, sensory disturbances (e.g. paresthesias such as electric shock sensations), anxiety, confusion, headache, lethargy, emotional lability, insomnia, hypomania, tinnitus, and seizures. While these events are generally self-limiting, there have been reports of serious discontinuation symptoms.
Patients should be monitored for these symptoms when discontinuing treatment with Effexor XR. A gradual reduction in the dose rather than abrupt cessation is recommended whenever possible. If intolerable symptoms occur following a decrease in the dose or upon discontinuation of treatment, then resuming the previously prescribed dose may be considered. Subsequently, the physician may continue decreasing the dose but at a more gradual rate (see DOSAGE AND ADMINISTRATION).
Insomnia and Nervousness
Treatment-emergent insomnia and nervousness were more commonly reported for patients treated with Effexor XR (venlafaxine hydrochloride) extended-release capsules than with placebo in pooled analyses of short-term major depressive disorder, GAD, Social Anxiety Disorder, and panic disorder studies, as shown in Table 5.
| Major Depressive Disorder |
GAD | Social Anxiety Disorder |
Panic Disorder |
|||||
| Symptom | Effexor XR n = 357 | Placebo n = 285 | Effexor XR n = 1381 | Placebo n = 555 | Effexor XR n = 819 | Placebo n = 695 | Effexor XR n = 1001 | Placebo n = 662 |
| Insomnia | 17% | 11% | 15% | 10% | 24% | 8% | 17% | 9% |
| Nervousness | 10% | 5% | 6% | 4% | 10% | 5% | 4% | 6% |
Insomnia and nervousness each led to drug discontinuation in 0.9% of the patients treated with Effexor XR in major depressive disorder studies.
In GAD trials, insomnia and nervousness led to drug discontinuation in 3% and 2%, respectively, of the patients treated with Effexor XR up to 8 weeks and 2% and 0.7%, respectively, of the patients treated with Effexor XR up to 6 months.
In Social Anxiety Disorder trials, insomnia and nervousness led to drug discontinuation in 2% and 1%, respectively, of the patients treated with Effexor XR up to 12 weeks and 2% and 3% respectively, of the patients treated with Effexor XR up to 6 months.
In panic disorder trials, insomnia and nervousness led to drug discontinuation in 1% and 0.1%, respectively, of the patients treated with Effexor XR up to 12 weeks.
Changes in Weight
Adult Patients: A loss of 5% or more of body weight occurred in 7% of Effexor XR-treated and 2% of placebo-treated patients in the short-term placebo-controlled major depressive disorder trials. The discontinuation rate for weight loss associated with Effexor XR was 0.1% in major depressive disorder studies. In placebo-controlled GAD studies, a loss of 7% or more of body weight occurred in 3% of Effexor XR patients and 1% of placebo patients who received treatment for up to 6 months. The discontinuation rate for weight loss was 0.3% for patients receiving Effexor XR in GAD studies for up to eight weeks. In placebo-controlled Social Anxiety Disorder trials, 4% of the Effexor XR-treated and 1% of the placebo-treated patients sustained a loss of 7% or more of body weight during up to 6 months of treatment. None of the patients receiving Effexor XR in Social Anxiety Disorder studies discontinued for weight loss. In placebo-controlled panic disorder trials, 3% of the Effexor XR-treated and 2% of the placebo-treated patients sustained a loss of 7% or more of body weight during up to 12 weeks of treatment. None of the patients receiving Effexor XR in panic disorder studies discontinued for weight loss.
The safety and efficacy of venlafaxine therapy in combination with weight loss agents, including phentermine, have not been established. Co-administration of Effexor XR and weight loss agents is not recommended. Effexor XR is not indicated for weight loss alone or in combination with other products.
Pediatric Patients: Weight loss has been observed in pediatric patients (ages 6-17) receiving Effexor XR. In a pooled analysis of four eight-week, double-blind, placebo-controlled, flexible dose outpatient trials for major depressive disorder (MDD) and generalized anxiety disorder (GAD), Effexor XR-treated patients lost an average of 0.45 kg (n = 333), while placebo-treated patients gained an average of 0.77 kg (n = 333). More patients treated with Effexor XR than with placebo experienced a weight loss of at least 3.5% in both the MDD and the GAD studies (18% of Effexor XR-treated patients vs. 3.6% of placebo-treated patients; p<0.001). In a 16-week, double-blind, placebo-controlled, flexible dose outpatient trial for Social Anxiety Disorder, Effexor XR-treated patients lost an average of 0.75 kg (n = 137), while placebo-treated patients gained an average of 0.76 kg (n = 148). More patients treated with Effexor XR than with placebo experienced a weight loss of at least 3.5% in the Social Anxiety Disorder study (47% of Effexor XR-treated patients vs. 14% of placebo-treated patients; p<0.001). Weight loss was not limited to patients with treatment-emergent anorexia (see PRECAUTIONS, General, Changes in Appetite).
The risks associated with longer-term Effexor XR use were assessed in an open-label MDD study of children and adolescents who received Effexor XR for up to six months. The children and adolescents in the study had increases in weight that were less than expected based on data from age- and sex-matched peers. The difference between observed weight gain and expected weight gain was larger for children (<12 years old) than for adolescents (≥12 years old).
Changes in Height
Pediatric Patients: During the eight-week, placebo-controlled GAD studies, Effexor XR-treated patients (ages 6-17) grew an average of 0.3 cm (n = 122), while placebo-treated patients grew an average of 1.0 cm (n = 132); p=0.041. This difference in height increase was most notable in patients younger than twelve. During the eight-week placebo-controlled MDD studies, Effexor XR-treated patients grew an average of 0.8 cm (n = 146), while placebo-treated patients grew an average of 0.7 cm (n = 147). During the 16-week, placebo-controlled Social Anxiety Disorder study, both the Effexor XR-treated (n = 109) and the placebo-treated (n = 112) patients each grew an average of 1.0 cm. In the six-month, open-label MDD study, children and adolescents had height increases that were less than expected based on data from age- and sex-matched peers. The difference between observed growth rates and expected growth rates was larger for children (<12 years old) than for adolescents (≥12 years old).
Changes in Appetite
Adult Patients: Treatment-emergent anorexia was more commonly reported for Effexor XR-treated (8%) than placebo-treated patients (4%) in the pool of short-term, double-blind, placebo-controlled major depressive disorder studies. The discontinuation rate for anorexia associated with Effexor XR was 1.0% in major depressive disorder studies. Treatment-emergent anorexia was more commonly reported for Effexor XR-treated (8%) than placebo-treated patients (2%) in the pool of short-term, double-blind, placebo-controlled GAD studies. The discontinuation rate for anorexia was 0.9% for patients receiving Effexor XR for up to 8 weeks in GAD studies. Treatment-emergent anorexia was more commonly reported for Effexor XR-treated (17%) than placebo-treated patients (2%) in the pool of short-term, double-blind, placebo-controlled Social Anxiety Disorder studies. The discontinuation rate for anorexia was 0.6% for patients receiving Effexor XR for up to 12 weeks in Social Anxiety Disorder studies; no patients discontinued for anorexia between week 12 and month 6. Treatment-emergent anorexia was more commonly reported for Effexor XR-treated (8%) than placebo-treated patients (3%) in the pool of short-term, double-blind, placebo-controlled panic disorder studies. The discontinuation rate for anorexia was 0.4% for patients receiving Effexor XR for up to 12 weeks in panic disorder studies.
Pediatric Patients: Decreased appetite has been observed in pediatric patients receiving Effexor XR. In the placebo-controlled trials for GAD and MDD, 10% of patients aged 6-17 treated with Effexor XR for up to eight weeks and 3% of patients treated with placebo reported treatment-emergent anorexia (decreased appetite). None of the patients receiving Effexor XR discontinued for anorexia or weight loss. In the placebo-controlled trial for Social Anxiety Disorder, 22% and 3% of patients aged 8-17 treated for up to 16 weeks with Effexor XR and placebo, respectively, reported treatment-emergent anorexia (decreased appetite). The discontinuation rates for anorexia were 0.7% and 0.0% for patients receiving Effexor XR and placebo, respectively; the discontinuation rates for weight loss were 0.7% for patients receiving either Effexor XR or placebo.
Activation of Mania/Hypomania
During premarketing major depressive disorder studies, mania or hypomania occurred in 0.3% of Effexor XR-treated patients and no placebo patients. In premarketing GAD studies, no Effexor XR-treated patients and 0.2% of placebo-treated patients experienced mania or hypomania. In premarketing Social Anxiety Disorder studies, 0.2% Effexor XR-treated patients and no placebo-treated patients experienced mania or hypomania. In premarketing panic disorder studies, 0.1% of Effexor XR-treated patients and no placebo-treated patients experienced mania or hypomania. In all premarketing major depressive disorder trials with Effexor (immediate release), mania or hypomania occurred in 0.5% of venlafaxine-treated patients compared with no placebo patients. Mania/hypomania has also been reported in a small proportion of patients with mood disorders who were treated with other marketed drugs to treat major depressive disorder. As with all drugs effective in the treatment of major depressive disorder, Effexor XR should be used cautiously in patients with a history of mania.
Hyponatremia
Hyponatremia may occur as a result of treatment with SSRIs and SNRIs, including Effexor XR. In many cases, this hyponatremia appears to be the result of the syndrome of inappropriate antidiuretic hormone secretion (SIADH). Cases with serum sodium lower than 110 mmol/L have been reported. Elderly patients may be at greater risk of developing hyponatremia with SSRIs and SNRIs. Also, patients taking diuretics or who are otherwise volume depleted may be at greater risk (see PRECAUTIONS, Geriatric Use). Discontinuation of Effexor XR should be considered in patients with symptomatic hyponatremia and appropriate medical intervention should be instituted.
Signs and symptoms of hyponatremia include headache, difficulty concentrating, memory impairment, confusion, weakness, and unsteadiness, which may lead to falls. Signs and symptoms associated with more severe and/or acute cases have included hallucination, syncope, seizure, coma, respiratory arrest, and death.
Seizures
During premarketing experience, no seizures occurred among 705 Effexor XR-treated patients in the major depressive disorder studies, among 1381 Effexor XR-treated patients in GAD studies, or among 819 Effexor XR-treated patients in Social Anxiety Disorder studies. In panic disorder studies, 1 seizure occurred among 1,001 Effexor XR-treated patients. In all premarketing major depressive disorder trials with Effexor (immediate release), seizures were reported at various doses in 0.3% (8/3082) of venlafaxine-treated patients. Effexor XR, like many antidepressants, should be used cautiously in patients with a history of seizures and should be discontinued in any patient who develops seizures.
Abnormal Bleeding
SSRIs and SNRIs, including Effexor XR, may increase the risk of bleeding events. Concomitant use of aspirin, nonsteroidal anti-inflammatory drugs, warfarin, and other anti-coagulants may add to this risk. Case reports and epidemiological studies (case-control and cohort design) have demonstrated an association between use of drugs that interfere with serotonin reuptake and the occurrence of gastrointestinal bleeding. Bleeding events related to SSRIs and SNRIs use have ranged from ecchymoses, hematomas, epistaxis, and petechiae to life-threatening hemorrhages.
Patients should be cautioned about the risk of bleeding associated with the concomitant use of Effexor XR and NSAIDs, aspirin, or other drugs that affect coagulation.
Serum Cholesterol Elevation
Clinically relevant increases in serum cholesterol were recorded in 5.3% of venlafaxine-treated patients and 0.0% of placebo-treated patients treated for at least 3 months in placebo-controlled trials (see ADVERSE REACTIONS-Laboratory Changes). Measurement of serum cholesterol levels should be considered during long-term treatment.
Interstitial Lung Disease and Eosinophilic Pneumonia
Interstitial lung disease and eosinophilic pneumonia associated with venlafaxine therapy have been rarely reported. The possibility of these adverse events should be considered in venlafaxine-treated patients who present with progressive dyspnea, cough or chest discomfort. Such patients should undergo a prompt medical evaluation, and discontinuation of venlafaxine therapy should be considered.
Use in Patients With Concomitant Illness
Premarketing experience with venlafaxine in patients with concomitant systemic illness is limited. Caution is advised in administering Effexor XR to patients with diseases or conditions that could affect hemodynamic responses or metabolism.
Venlafaxine has not been evaluated or used to any appreciable extent in patients with a recent history of myocardial infarction or unstable heart disease. Patients with these diagnoses were systematically excluded from many clinical studies during venlafaxine's premarketing testing. The electrocardiograms were analyzed for 275 patients who received Effexor XR and 220 patients who received placebo in 8- to 12-week double-blind, placebo-controlled trials in major depressive disorder, for 610 patients who received Effexor XR and 298 patients who received placebo in 8-week double-blind, placebo-controlled trials in GAD, for 593 patients who received Effexor XR and 534 patients who received placebo in 12-week double-blind, placebo-controlled trials in Social Anxiety Disorder, and for 661 patients who received Effexor XR and 395 patients who received placebo in three 10- to 12-week double-blind, placebo-controlled trials in panic disorder. The mean change from baseline in corrected QT interval (QTc) for Effexor XR-treated patients in major depressive disorder studies was increased relative to that for placebo-treated patients (increase of 4.7 msec for Effexor XR and decrease of 1.9 msec for placebo). The mean change from baseline in QTc interval for Effexor XR-treated patients in the GAD studies did not differ significantly from that with placebo. The mean change from baseline in QTc interval for Effexor XR-treated patients in the Social Anxiety Disorder studies was increased relative to that for placebo-treated patients (increase of 3.4 msec for Effexor XR and decrease of 1.6 msec for placebo). The mean change from baseline in QTc interval for Effexor XR-treated patients in the panic disorder studies was increased relative to that for placebo-treated patients (increase of 1.5 msec for Effexor XR and decrease of 0.7 msec for placebo).
In these same trials, the mean change from baseline in heart rate for Effexor XR-treated patients in the major depressive disorder studies was significantly higher than that for placebo (a mean increase of 4 beats per minute for Effexor XR and 1 beat per minute for placebo). The mean change from baseline in heart rate for Effexor XR-treated patients in the GAD studies was significantly higher than that for placebo (a mean increase of 3 beats per minute for Effexor XR and no change for placebo). The mean change from baseline in heart rate for Effexor XR-treated patients in the Social Anxiety Disorder studies was significantly higher than that for placebo (a mean increase of 5 beats per minute for Effexor XR and no change for placebo). The mean change from baseline in heart rate for Effexor XR-treated patients in the panic disorder studies was significantly higher than that for placebo (a mean increase of 3 beats per minute for Effexor XR and a mean decrease of less than 1 beat per minute for placebo).
In a flexible-dose study, with Effexor (immediate release) doses in the range of 200 to 375 mg/day and mean dose greater than 300 mg/day, Effexor-treated patients had a mean increase in heart rate of 8.5 beats per minute compared with 1.7 beats per minute in the placebo group.
As increases in heart rate were observed, caution should be exercised in patients whose underlying medical conditions might be compromised by increases in heart rate (eg, patients with hyperthyroidism, heart failure, or recent myocardial infarction).
Evaluation of the electrocardiograms for 769 patients who received Effexor (immediate release) in 4- to 6-week double-blind, placebo-controlled trials showed that the incidence of trial-emergent conduction abnormalities did not differ from that with placebo.
In patients with renal impairment (GFR = 10 to 70 mL/min) or cirrhosis of the liver, the clearances of venlafaxine and its active metabolites were decreased, thus prolonging the elimination half-lives of these substances. A lower dose may be necessary (see DOSAGE AND ADMINISTRATION). Effexor XR, like all drugs effective in the treatment of major depressive disorder, should be used with caution in such patients.
Information for Patients
Prescribers or other health professionals should inform patients, their families, and their caregivers about the benefits and risks associated with treatment with Effexor XR and should counsel them in its appropriate use. A patient Medication Guide about “Antidepressant Medicines, Depression and Other Serious Mental Illness, and Suicidal Thoughts or Actions” is available for Effexor XR. The prescriber or health professional should instruct patients, their families, and their caregivers to read the Medication Guide and should assist them in understanding its contents. Patients should be given the opportunity to discuss the contents of the Medication Guide and to obtain answers to any questions they may have. The complete text of the Medication Guide is reprinted at the end of this document.
Patients should be advised of the following issues and asked to alert their prescriber if these occur while taking Effexor XR.
Clinical Worsening and Suicide Risk: Patients, their families, and their caregivers should be encouraged to be alert to the emergence of anxiety, agitation, panic attacks, insomnia, irritability, hostility, aggressiveness, impulsivity, akathisia (psychomotor restlessness), hypomania, mania, other unusual changes in behavior, worsening of depression, and suicidal ideation, especially early during antidepressant treatment and when the dose is adjusted up or down. Families and caregivers of patients should be advised to look for the emergence of such symptoms on a day-to-day basis, since changes may be abrupt. Such symptoms should be reported to the patient's prescriber or health professional, especially if they are severe, abrupt in onset, or were not part of the patient's presenting symptoms. Symptoms such as these may be associated with an increased risk for suicidal thinking and behavior and indicate a need for very close monitoring and possibly changes in the medication.
Interference with Cognitive and Motor Performance
Clinical studies were performed to examine the effects of venlafaxine on behavioral performance of healthy individuals. The results revealed no clinically significant impairment of psychomotor, cognitive, or complex behavior performance. However, since any psychoactive drug may impair judgment, thinking, or motor skills, patients should be cautioned about operating hazardous machinery, including automobiles, until they are reasonably certain that venlafaxine therapy does not adversely affect their ability to engage in such activities.
Concomitant Medication
Patients should be advised to inform their physicians if they are taking, or plan to take, any prescription or over-the-counter drugs, including herbal preparations and nutritional supplements, since there is a potential for interactions.
Patients should be cautioned about the risk of serotonin syndrome with the concomitant use of Effexor XR and triptans, tramadol, tryptophan supplements or other serotonergic agents (see WARNINGS, Serotonin Syndrome and PRECAUTIONS, Drug Interactions, CNS-Active Drugs).
Patients should be cautioned about the concomitant use of Effexor XR and NSAIDs, aspirin, warfarin, or other drugs that affect coagulation since combined use of psychotropic drugs that interfere with serotonin reuptake and these agents has been associated with an increased risk of bleeding (see PRECAUTIONS, Abnormal Bleeding).
Alcohol
Although venlafaxine has not been shown to increase the impairment of mental and motor skills caused by alcohol, patients should be advised to avoid alcohol while taking venlafaxine.
Allergic Reactions
Patients should be advised to notify their physician if they develop a rash, hives, or a related allergic phenomenon.
Pregnancy
Patients should be advised to notify their physician if they become pregnant or intend to become pregnant during therapy.
Nursing
Patients should be advised to notify their physician if they are breast-feeding an infant.
Mydriasis
Mydriasis (prolonged dilation of the pupils of the eye) has been reported with venlafaxine. Patients should be advised to notify their physician if they have a history of glaucoma or a history of increased intraocular pressure (see WARNINGS).
Laboratory Tests
There are no specific laboratory tests recommended.
Drug Interactions
As with all drugs, the potential for interaction by a variety of mechanisms is a possibility.
Alcohol
A single dose of ethanol (0.5 g/kg) had no effect on the pharmacokinetics of venlafaxine or O-desmethylvenlafaxine (ODV) when venlafaxine was administered at 150 mg/day in 15 healthy male subjects. Additionally, administration of venlafaxine in a stable regimen did not exaggerate the psychomotor and psychometric effects induced by ethanol in these same subjects when they were not receiving venlafaxine.
Cimetidine
Concomitant administration of cimetidine and venlafaxine in a steady-state study for both drugs resulted in inhibition of first-pass metabolism of venlafaxine in 18 healthy subjects. The oral clearance of venlafaxine was reduced by about 43%, and the exposure (AUC) and maximum concentration (Cmax) of the drug were increased by about 60%. However, coadministration of cimetidine had no apparent effect on the pharmacokinetics of ODV, which is present in much greater quantity in the circulation than venlafaxine. The overall pharmacological activity of venlafaxine plus ODV is expected to increase only slightly, and no dosage adjustment should be necessary for most normal adults. However, for patients with pre-existing hypertension, and for elderly patients or patients with hepatic dysfunction, the interaction associated with the concomitant use of venlafaxine and cimetidine is not known and potentially could be more pronounced. Therefore, caution is advised with such patients.
Diazepam
Under steady-state conditions for venlafaxine administered at 150 mg/day, a single 10 mg dose of diazepam did not appear to affect the pharmacokinetics of either venlafaxine or ODV in 18 healthy male subjects. Venlafaxine also did not have any effect on the pharmacokinetics of diazepam or its active metabolite, desmethyldiazepam, or affect the psychomotor and psychometric effects induced by diazepam.
Haloperidol
Venlafaxine administered under steady-state conditions at 150 mg/day in 24 healthy subjects decreased total oral-dose clearance (Cl/F) of a single 2 mg dose of haloperidol by 42%, which resulted in a 70% increase in haloperidol AUC. In addition, the haloperidol Cmax increased 88% when coadministered with venlafaxine, but the haloperidol elimination half-life (t1/2) was unchanged. The mechanism explaining this finding is unknown.
Lithium
The steady-state pharmacokinetics of venlafaxine administered at 150 mg/day were not affected when a single 600 mg oral dose of lithium was administered to 12 healthy male subjects. ODV also was unaffected. Venlafaxine had no effect on the pharmacokinetics of lithium (see also CNS-Active Drugs, below).
Drugs Highly Bound to Plasma Proteins
Venlafaxine is not highly bound to plasma proteins; therefore, administration of Effexor XR to a patient taking another drug that is highly protein bound should not cause increased free concentrations of the other drug.
Drugs that Interfere with Hemostasis (e.g., NSAIDs, Aspirin, and Warfarin)
Serotonin release by platelets plays an important role in hemostasis. Epidemiological studies of the case-control and cohort design that have demonstrated an association between use of psychotropic drugs that interfere with serotonin reuptake and the occurrence of upper gastrointestinal bleeding have also shown that concurrent use of an NSAID or aspirin may potentiate this risk of bleeding. Altered anticoagulant effects, including increased bleeding, have been reported when SSRIs and SNRIs are coadministered with warfarin. Patients receiving warfarin therapy should be carefully monitored when Effexor XR is initiated or discontinued.
Drugs that Inhibit Cytochrome P450 Isoenzymes
CYP2D6 Inhibitors: In vitro and in vivo studies indicate that venlafaxine is metabolized to its active metabolite, ODV, by CYP2D6, the isoenzyme that is responsible for the genetic polymorphism seen in the metabolism of many antidepressants. Therefore, the potential exists for a drug interaction between drugs that inhibit CYP2D6-mediated metabolism of venlafaxine, reducing the metabolism of venlafaxine to ODV, resulting in increased plasma concentrations of venlafaxine and decreased concentrations of the active metabolite. CYP2D6 inhibitors such as quinidine would be expected to do this, but the effect would be similar to what is seen in patients who are genetically CYP2D6 poor metabolizers (see Metabolism and Excretion under CLINICAL PHARMACOLOGY). Therefore, no dosage adjustment is required when venlafaxine is coadministered with a CYP2D6 inhibitor.
Ketoconazole: A pharmacokinetic study with ketoconazole 100 mg b.i.d. with a single dose of venlafaxine 50 mg in extensive metabolizers (EM; n = 14) and 25 mg in poor metabolizers (PM; n = 6) of CYP2D6 resulted in higher plasma concentrations of both venlafaxine and O-desmethylvenlafaxine (ODV) in most subjects following administration of ketoconazole. Venlafaxine Cmax increased by 26% in EM subjects and 48% in PM subjects. Cmax values for ODV increased by 14% and 29% in EM and PM subjects, respectively.
Venlafaxine AUC increased by 21% in EM subjects and 70% in PM subjects (range in PMs -2% to 206%), and AUC values for ODV increased by 23% and 33% in EM and PM (range in PMs -38% to 105%) subjects, respectively. Combined AUCs of venlafaxine and ODV increased on average by approximately 23% in EMs and 53% in PMs (range in PMs 4% to 134%).
Concomitant use of CYP3A4 inhibitors and venlafaxine may increase levels of venlafaxine and ODV. Therefore, caution is advised if a patient's therapy includes a CYP3A4 inhibitor and venlafaxine concomitantly.
Drugs Metabolized by Cytochrome P450 Isoenzymes
CYP2D6: In vitro studies indicate that venlafaxine is a relatively weak inhibitor of CYP2D6. These findings have been confirmed in a clinical drug interaction study comparing the effect of venlafaxine with that of fluoxetine on the CYP2D6-mediated metabolism of dextromethorphan to dextrorphan.
Imipramine - Venlafaxine did not affect the pharmacokinetics of imipramine and 2-OH-imipramine. However, desipramine AUC, Cmax, and Cmin increased by about 35% in the presence of venlafaxine. The 2-OH-desipramine AUC's increased by at least 2.5 fold (with venlafaxine 37.5 mg q12h) and by 4.5 fold (with venlafaxine 75 mg q12h). Imipramine did not affect the pharmacokinetics of venlafaxine and ODV. The clinical significance of elevated 2-OH-desipramine levels is unknown.
Metoprolol - Concomitant administration of venlafaxine (50 mg every 8 hours for 5 days) and metoprolol (100 mg every 24 hours for 5 days) to 18 healthy male subjects in a pharmacokinetic interaction study for both drugs resulted in an increase of plasma concentrations of metoprolol by approximately 30-40% without altering the plasma concentrations of its active metabolite, α-hydroxymetoprolol. Metoprolol did not alter the pharmacokinetic profile of venlafaxine or its active metabolite, O-desmethylvenlafaxine.
Venlafaxine appeared to reduce the blood pressure lowering effect of metoprolol in this study. The clinical relevance of this finding for hypertensive patients is unknown. Caution should be exercised with co-administration of venlafaxine and metoprolol.
Venlafaxine treatment has been associated with dose-related increases in blood pressure in some patients. It is recommended that patients receiving Effexor XR have regular monitoring of blood pressure (see WARNINGS).
Risperidone - Venlafaxine administered under steady-state conditions at 150 mg/day slightly inhibited the CYP2D6-mediated metabolism of risperidone (administered as a single 1 mg oral dose) to its active metabolite, 9-hydroxyrisperidone, resulting in an approximate 32% increase in risperidone AUC. However, venlafaxine coadministration did not significantly alter the pharmacokinetic profile of the total active moiety (risperidone plus 9-hydroxyrisperidone).
CYP3A4: Venlafaxine did not inhibit CYP3A4 in vitro. This finding was confirmed in vivo by clinical drug interaction studies in which venlafaxine did not inhibit the metabolism of several CYP3A4 substrates, including alprazolam, diazepam, and terfenadine.
Indinavir - In a study of 9 healthy volunteers, venlafaxine administered under steady-state conditions at 150 mg/day resulted in a 28% decrease in the AUC of a single 800 mg oral dose of indinavir and a 36% decrease in indinavir Cmax. Indinavir did not affect the pharmacokinetics of venlafaxine and ODV. The clinical significance of this finding is unknown.
CYP1A2: Venlafaxine did not inhibit CYP1A2 in vitro. This finding was confirmed in vivo by a clinical drug interaction study in which venlafaxine did not inhibit the metabolism of caffeine, a CYP1A2 substrate.
CYP2C9: Venlafaxine did not inhibit CYP2C9 in vitro. In vivo, venlafaxine 75 mg by mouth every 12 hours did not alter the pharmacokinetics of a single 500 mg dose of tolbutamide or the CYP2C9 mediated formation of 4-hydroxy-tolbutamide.
CYP2C19: Venlafaxine did not inhibit the metabolism of diazepam, which is partially metabolized by CYP2C19 (see Diazepam above).
Monoamine Oxidase Inhibitors
See CONTRAINDICATIONS and WARNINGS.
CNS-Active Drugs
The risk of using venlafaxine in combination with other CNS-active drugs has not been systematically evaluated (except in the case of those CNS-active drugs noted above). Consequently, caution is advised if the concomitant administration of venlafaxine and such drugs is required.
Serotonergic Drugs: Based on the mechanism of action of Effexor XR and the potential for serotonin syndrome, caution is advised when Effexor XR is co-administered with other drugs that may affect the serotonergic neurotransmitter systems, such as triptans, SSRIs, other SNRIs, linezolid (an antibiotic which is a reversible non-selective MAOI), lithium, tramadol, or St. John's Wort (see WARNINGS, Serotonin Syndrome). If concomitant treatment of Effexor XR with these drugs is clinically warranted, careful observation of the patient is advised, particularly during treatment initiation and dose increases (see WARNINGS, Serotonin Syndrome). The concomitant use of Effexor XR with tryptophan supplements is not recommended (see WARNINGS, Serotonin Syndrome).
Triptans: There have been rare postmarketing reports of serotonin syndrome with use of an SSRI and a triptan. If concomitant treatment of Effexor XR with a triptan is clinically warranted, careful observation of the patient is advised, particularly during treatment initiation and dose increases (see WARNINGS, Serotonin Syndrome).
Electroconvulsive Therapy
There are no clinical data establishing the benefit of electroconvulsive therapy combined with Effexor XR (venlafaxine hydrochloride) extended-release capsules treatment.
Postmarketing Spontaneous Drug Interaction Reports
See ADVERSE REACTIONS, Postmarketing Reports.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Venlafaxine was given by oral gavage to mice for 18 months at doses up to 120 mg/kg per day, which was 1.7 times the maximum recommended human dose on a mg/m2 basis. Venlafaxine was also given to rats by oral gavage for 24 months at doses up to 120 mg/kg per day. In rats receiving the 120 mg/kg dose, plasma concentrations of venlafaxine at necropsy were 1 times (male rats) and 6 times (female rats) the plasma concentrations of patients receiving the maximum recommended human dose. Plasma levels of the O-desmethyl metabolite were lower in rats than in patients receiving the maximum recommended dose. Tumors were not increased by venlafaxine treatment in mice or rats.
Mutagenesis
Venlafaxine and the major human metabolite, O-desmethylvenlafaxine (ODV), were not mutagenic in the Ames reverse mutation assay in Salmonella bacteria or the Chinese hamster ovary/HGPRT mammalian cell forward gene mutation assay. Venlafaxine was also not mutagenic or clastogenic in the in vitro BALB/c-3T3 mouse cell transformation assay, the sister chromatid exchange assay in cultured Chinese hamster ovary cells, or in the in vivo chromosomal aberration assay in rat bone marrow. ODV was not clastogenic in the in vitro Chinese hamster ovary cell chromosomal aberration assay, but elicited a clastogenic response in the in vivo chromosomal aberration assay in rat bone marrow.
Impairment of Fertility
Reproduction and fertility studies in rats showed no effects on male or female fertility at oral doses of up to 2 times the maximum recommended human dose on a mg/m2 basis.
Pregnancy
Teratogenic Effects - Pregnancy Category C
Venlafaxine did not cause malformations in offspring of rats or rabbits given doses up to 2.5 times (rat) or 4 times (rabbit) the maximum recommended human daily dose on a mg/m2 basis. However, in rats, there was a decrease in pup weight, an increase in stillborn pups, and an increase in pup deaths during the first 5 days of lactation, when dosing began during pregnancy and continued until weaning. The cause of these deaths is not known. These effects occurred at 2.5 times (mg/m2) the maximum human daily dose. The no effect dose for rat pup mortality was 0.25 times the human dose on a mg/m2 basis. There are no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Non-teratogenic Effects
Neonates exposed to Effexor XR, other SNRIs (Serotonin and Norepinephrine Reuptake Inhibitors), or SSRIs (Selective Serotonin Reuptake Inhibitors), late in the third trimester have developed complications requiring prolonged hospitalization, respiratory support, and tube feeding. Such complications can arise immediately upon delivery. Reported clinical findings have included respiratory distress, cyanosis, apnea, seizures, temperature instability, feeding difficulty, vomiting, hypoglycemia, hypotonia, hypertonia, hyperreflexia, tremor, jitteriness, irritability, and constant crying. These features are consistent with either a direct toxic effect of SSRIs and SNRIs or, possibly, a drug discontinuation syndrome. It should be noted that, in some cases, the clinical picture is consistent with serotonin syndrome (see PRECAUTIONS-Drug Interactions-CNS-Active Drugs). When treating a pregnant woman with Effexor XR during the third trimester, the physician should carefully consider the potential risks and benefits of treatment (see DOSAGE AND ADMINISTRATION).
Labor and Delivery
The effect of venlafaxine on labor and delivery in humans is unknown.
Nursing Mothers
Venlafaxine and ODV have been reported to be excreted in human milk. Because of the potential for serious adverse reactions in nursing infants from Effexor XR, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
Safety and effectiveness in the pediatric population have not been established (see BOX WARNING and WARNINGS, Clinical Worsening and Suicide Risk). Two placebo-controlled trials in 766 pediatric patients with MDD and two placebo-controlled trials in 793 pediatric patients with GAD have been conducted with Effexor XR, and the data were not sufficient to support a claim for use in pediatric patients.
Anyone considering the use of Effexor XR in a child or adolescent must balance the potential risks with the clinical need.
Although no studies have been designed to primarily assess Effexor XR's impact on the growth, development, and maturation of children and adolescents, the studies that have been done suggest that Effexor XR may adversely affect weight and height (see PRECAUTIONS, General, Changes in Height and Changes in Weight ). Should the decision be made to treat a pediatric patient with Effexor XR, regular monitoring of weight and height is recommended during treatment, particularly if it is to be continued long term. The safety of Effexor XR treatment for pediatric patients has not been systematically assessed for chronic treatment longer than six months in duration.
In the studies conducted in pediatric patients (ages 6-17), the occurrence of blood pressure and cholesterol increases considered to be clinically relevant in pediatric patients was similar to that observed in adult patients. Consequently, the precautions for adults apply to pediatric patients (see WARNINGS, Sustained Hypertension, and PRECAUTIONS, General, Serum Cholesterol Elevation).
Geriatric Use
Approximately 4% (14/357), 6% (77/1381), 1% (10/819), and 2% (16/1001) of Effexor XR-treated patients in placebo-controlled premarketing major depressive disorder, GAD, Social Anxiety Disorder trials, and panic disorder trials, respectively, were 65 years of age or over. Of 2,897 Effexor-treated (immediate release) patients in premarketing phase major depressive disorder studies, 12% (357) were 65 years of age or over. No overall differences in effectiveness or safety were observed between geriatric patients and younger patients, and other reported clinical experience generally has not identified differences in response between the elderly and younger patients. However, greater sensitivity of some older individuals cannot be ruled out. SSRIs and SNRIs, including Effexor XR have been associated with cases of clinically significant hyponatremia in elderly patients, who may be at greater risk for this adverse event (see PRECAUTIONS, Hyponatremia).
The pharmacokinetics of venlafaxine and ODV are not substantially altered in the elderly (see CLINICAL PHARMACOLOGY). No dose adjustment is recommended for the elderly on the basis of age alone, although other clinical circumstances, some of which may be more common in the elderly, such as renal or hepatic impairment, may warrant a dose reduction (see DOSAGE AND ADMINISTRATION).
ADVERSE REACTIONS
The information included in the Adverse Findings Observed in Short-Term, Placebo-Controlled Studies with Effexor XR subsection is based on data from a pool of three 8- and 12-week controlled clinical trials in major depressive disorder (includes two U.S. trials and one European trial), on data up to 8 weeks from a pool of five controlled clinical trials in GAD with Effexor XR®, on data up to 12 weeks from a pool of five controlled clinical trials in Social Anxiety Disorder, and on data up to 12 weeks from a pool of four controlled clinical trials in panic disorder. Information on additional adverse events associated with Effexor XR in the entire development program for the formulation and with Effexor (immediate release) is included in the Other Adverse Events Observed During the Premarketing Evaluation of Effexor and Effexor XR subsection (see also WARNINGS and PRECAUTIONS).
Adverse Findings Observed in Short-Term, Placebo-Controlled Studies with Effexor XR
Adverse Events Associated with Discontinuation of Treatment
Approximately 11% of the 357 patients who received Effexor XR® (venlafaxine hydrochloride) extended-release capsules in placebo-controlled clinical trials for major depressive disorder discontinued treatment due to an adverse experience, compared with 6% of the 285 placebo-treated patients in those studies. Approximately 18% of the 1381 patients who received Effexor XR capsules in placebo-controlled clinical trials for GAD discontinued treatment due to an adverse experience, compared with 12% of the 555 placebo-treated patients in those studies. Approximately 15% of the 819 patients who received Effexor XR capsules in placebo-controlled clinical trials for Social Anxiety Disorder discontinued treatment due to an adverse experience, compared with 5% of the 695 placebo-treated patients in those studies. Approximately 7% of the 1,001 patients who received Effexor XR capsules in placebo-controlled clinical trials for panic disorder discontinued treatment due to an adverse experience, compared with 6% of the 662 placebo-treated patients in those studies. The most common events leading to discontinuation and considered to be drug-related (ie, leading to discontinuation in at least 1% of the Effexor XR-treated patients at a rate at least twice that of placebo for any indication) are shown in Table 6.
Adverse Events Occurring at an Incidence of 2% or More Among Effexor XR-Treated Patients
Tables 7, 8, 9, and 10 enumerate the incidence, rounded to the nearest percent, of treatment-emergent adverse events that occurred during acute therapy of major depressive disorder (up to 12 weeks; dose range of 75 to 225 mg/day), of GAD (up to 8 weeks; dose range of 37.5 to 225 mg/day), of Social Anxiety Disorder (up to 12 weeks; dose range of 75 to 225 mg/day), and of panic disorder (up to 12 weeks; dose range of 37.5 to 225 mg/day), respectively, in 2% or more of patients treated with Effexor XR (venlafaxine hydrochloride) where the incidence in patients treated with Effexor XR was greater than the incidence for the respective placebo-treated patients. The table shows the percentage of patients in each group who had at least one episode of an event at some time during their treatment. Reported adverse events were classified using a standard COSTART-based Dictionary terminology.
The prescriber should be aware that these figures cannot be used to predict the incidence of side effects in the course of usual medical practice where patient characteristics and other factors differ from those which prevailed in the clinical trials. Similarly, the cited frequencies cannot be compared with figures obtained from other clinical investigations involving different treatments, uses and investigators. The cited figures, however, do provide the prescribing physician with some basis for estimating the relative contribution of drug and nondrug factors to the side effect incidence rate in the population studied.
Commonly Observed Adverse Events from Tables 7, 8, 9, and 10:
Major Depressive Disorder
Note in particular the following adverse events that occurred in at least 5% of the Effexor XR patients and at a rate at least twice that of the placebo group for all placebo-controlled trials for the major depressive disorder indication (Table 7): Abnormal ejaculation, gastrointestinal complaints (nausea, dry mouth, and anorexia), CNS complaints (dizziness, somnolence, and abnormal dreams), and sweating. In the two U.S. placebo-controlled trials, the following additional events occurred in at least 5% of Effexor XR-treated patients (n = 192) and at a rate at least twice that of the placebo group: Abnormalities of sexual function (impotence in men, anorgasmia in women, and libido decreased), gastrointestinal complaints (constipation and flatulence), CNS complaints (insomnia, nervousness, and tremor), problems of special senses (abnormal vision), cardiovascular effects (hypertension and vasodilatation), and yawning.
Generalized Anxiety Disorder
Note in particular the following adverse events that occurred in at least 5% of the Effexor XR patients and at a rate at least twice that of the placebo group for all placebo-controlled trials for the GAD indication (Table 8): Abnormalities of sexual function (abnormal ejaculation and impotence), gastrointestinal complaints (nausea, dry mouth, anorexia, and constipation), problems of special senses (abnormal vision), and sweating.
Social Anxiety Disorder
Note in particular the following adverse events that occurred in at least 5% of the Effexor XR patients and at a rate at least twice that of the placebo group for the 5 placebo-controlled trials for the Social Anxiety Disorder indication (Table 9): Asthenia, gastrointestinal complaints (anorexia, constipation, dry mouth, nausea), CNS complaints (insomnia, libido decreased, nervousness, somnolence, tremor), abnormalities of sexual function (abnormal ejaculation, impotence), yawn, and sweating.
In the 6-month trial, the following adverse events occurred twice as often in the 150-225 mg/day Effexor XR group compared to the 75 mg/day Effexor XR group and placebo: vasodilation, libido decreased, tremor, yawn, abnormal vision, and impotence.
Panic Disorder
Note in particular the following adverse events that occurred in at least 5% of the Effexor XR patients and at a rate at least twice that of the placebo group for 4 placebo-controlled trials for the panic disorder indication (Table 10): gastrointestinal complaints (anorexia, constipation, dry mouth), CNS complaints (somnolence, tremor), abnormalities of sexual function (abnormal ejaculation), and sweating.
Vital Sign Changes
Effexor XR (venlafaxine hydrochloride) extended-release capsules treatment for up to 12 weeks in premarketing placebo-controlled major depressive disorder trials was associated with a mean final on-therapy increase in pulse rate of approximately 2 beats per minute, compared with 1 beat per minute for placebo. Effexor XR treatment for up to 8 weeks in premarketing placebo-controlled GAD trials was associated with a mean final on-therapy increase in pulse rate of approximately 2 beats per minute, compared with less than 1 beat per minute for placebo. Effexor XR treatment for up to 12 weeks in premarketing placebo-controlled Social Anxiety Disorder trials was associated with a mean final on-therapy increase in pulse rate of approximately 3 beats per minute, compared with an increase of 1 beat per minute for placebo. Effexor XR treatment for up to 12 weeks in premarketing placebo-controlled panic disorder trials was associated with a mean final on-therapy increase in pulse rate of approximately 1 beat per minute, compared with a decrease of less than 1 beat per minute for placebo. (See the Sustained Hypertension and Elevations in Systolic and Diastolic Blood Pressure sections of WARNINGS for effects on blood pressure.)
In a flexible-dose study, with Effexor (immediate release) doses in the range of 200 to 375 mg/day and mean dose greater than 300 mg/day, the mean pulse was increased by about 2 beats per minute compared with a decrease of about 1 beat per minute for placebo.
Laboratory Changes
Serum Cholesterol
Effexor XR (venlafaxine hydrochloride) extended-release capsules treatment for up to 12 weeks in premarketing placebo-controlled trials for major depressive disorder was associated with a mean final on-therapy increase in serum cholesterol concentration of approximately 1.5 mg/dL compared with a mean final decrease of 7.4 mg/dL for placebo. Effexor XR treatment for up to 8 weeks and up to 6 months in premarketing placebo-controlled GAD trials was associated with mean final on-therapy increases in serum cholesterol concentration of approximately 1.0 mg/dL and 2.3 mg/dL, respectively while placebo subjects experienced mean final decreases of 4.9 mg/dL and 7.7 mg/dL, respectively. Effexor XR treatment for up to 12 weeks and up to 6 months in premarketing placebo-controlled Social Anxiety Disorder trials was associated with mean final on-therapy increases in serum cholesterol concentration of approximately 7.9 mg/dL and 5.6 mg/dL, respectively, compared with mean final decreases of 2.9 and 4.2 mg/dL, respectively, for placebo. Effexor XR treatment for up to 12 weeks in premarketing placebo-controlled panic disorder trials was associated with mean final on-therapy increases in serum cholesterol concentration of approximately 5.8 mg/dL compared with a mean final decrease of 3.7 mg/dL for placebo.
Patients treated with Effexor (immediate release) for at least 3 months in placebo-controlled 12-month extension trials had a mean final on-therapy increase in total cholesterol of 9.1 mg/dL compared with a decrease of 7.1 mg/dL among placebo-treated patients. This increase was duration dependent over the study period and tended to be greater with higher doses. Clinically relevant increases in serum cholesterol, defined as 1) a final on-therapy increase in serum cholesterol ≥50 mg/dL from baseline and to a value ≥261 mg/dL, or 2) an average on-therapy increase in serum cholesterol ≥50 mg/dL from baseline and to a value ≥261 mg/dL, were recorded in 5.3% of venlafaxine-treated patients and 0.0% of placebo-treated patients (see PRECAUTIONS-General-Serum Cholesterol Elevation).
Serum Triglycerides
Effexor XR treatment for up to 12 weeks in pooled premarketing Social Anxiety Disorder trials was associated with a mean final on-therapy increase in fasting serum triglyceride concentration of approximately 8.2 mg/dL, compared with a mean final increase of 0.4 mg/dL for placebo. Effexor XR treatment for up to 6 months in a premarketing Social Anxiety Disorder trial was associated with a mean final on-therapy increase in fasting serum triglyceride concentration of approximately 11.8 mg/dL, compared with a mean final on-therapy increase of 1.8 mg/dL for placebo.
Effexor XR treatment for up to 12 weeks in pooled premarketing Panic Disorder trials was associated with a mean final on-therapy increase in fasting serum triglyceride concentration of approximately 5.9 mg/dL, compared with a mean final increase of 0.9 mg/dL for placebo. Effexor XR treatment for up to 6 months in a premarketing Panic Disorder trial was associated with a mean final on-therapy increase in fasting serum triglyceride concentration of approximately 9.3 mg/dL, compared with a mean final on-therapy decrease of 0.3 mg/dL for placebo.
ECG Changes
In a flexible-dose study, with Effexor (immediate release) doses in the range of 200 to 375 mg/day and mean dose greater than 300 mg/day, the mean change in heart rate was 8.5 beats per minute compared with 1.7 beats per minute for placebo.
(See the Use in Patients with Concomitant Illness section of PRECAUTIONS.)
Other Adverse Events Observed During the Premarketing Evaluation of Effexor and Effexor XR
During its premarketing assessment, multiple doses of Effexor XR were administered to 705 patients in Phase 3 major depressive disorder studies and Effexor was administered to 96 patients. During its premarketing assessment, multiple doses of Effexor XR were also administered to 1381 patients in Phase 3 GAD studies, 819 patients in Phase 3 Social Anxiety Disorder studies, and 1314 patients in Phase 3 panic disorder studies. In addition, in premarketing assessment of Effexor, multiple doses were administered to 2897 patients in Phase 2 to Phase 3 studies for major depressive disorder. The conditions and duration of exposure to venlafaxine in both development programs varied greatly, and included (in overlapping categories) open and double-blind studies, uncontrolled and controlled studies, inpatient (Effexor only) and outpatient studies, fixed-dose, and titration studies. Untoward events associated with this exposure were recorded by clinical investigators using terminology of their own choosing. Consequently, it is not possible to provide a meaningful estimate of the proportion of individuals experiencing adverse events without first grouping similar types of untoward events into a smaller number of standardized event categories.
In the tabulations that follow, reported adverse events were classified using a standard COSTART-based Dictionary terminology. The frequencies presented, therefore, represent the proportion of the 7212 patients exposed to multiple doses of either formulation of venlafaxine who experienced an event of the type cited on at least one occasion while receiving venlafaxine. All reported events are included except those already listed in Tables 7, 8, 9, and 10 and those events for which a drug cause was remote. If the COSTART term for an event was so general as to be uninformative, it was replaced with a more informative term. It is important to emphasize that, although the events reported occurred during treatment with venlafaxine, they were not necessarily caused by it.
Events are further categorized by body system and listed in order of decreasing frequency using the following definitions: frequent adverse events are defined as those occurring on one or more occasions in at least 1/100 patients; infrequent adverse events are those occurring in 1/100 to 1/1000 patients; rare events are those occurring in fewer than 1/1000 patients.
Body as a whole - Frequent: chest pain substernal, chills, fever, neck pain; Infrequent: face edema, intentional injury, malaise, moniliasis, neck rigidity, pelvic pain, photosensitivity reaction, suicide attempt, withdrawal syndrome; Rare: appendicitis, bacteremia, carcinoma, cellulitis, granuloma.
Cardiovascular system - Frequent: migraine, tachycardia; Infrequent: angina pectoris, arrhythmia, bradycardia, extrasystoles, hypotension, peripheral vascular disorder (mainly cold feet and/or cold hands), postural hypotension, syncope; Rare: aortic aneurysm, arteritis, first-degree atrioventricular block, bigeminy, bundle branch block, capillary fragility, cerebral ischemia, coronary artery disease, congestive heart failure, heart arrest, hematoma, cardiovascular disorder (mitral valve and circulatory disturbance), mucocutaneous hemorrhage, myocardial infarct, pallor, sinus arrhythmia, thrombophlebitis.
Digestive system - Frequent: increased appetite; Infrequent: bruxism, colitis, dysphagia, tongue edema, eructation, esophagitis, gastritis, gastroenteritis, gastrointestinal ulcer, gingivitis, glossitis, rectal hemorrhage, hemorrhoids, melena, oral moniliasis, stomatitis, mouth ulceration; Rare: abdominal distension, biliary pain, cheilitis, cholecystitis, cholelithiasis, esophageal spasms, duodenitis, hematemesis, gastroesophageal reflux disease, gastrointestinal hemorrhage, gum hemorrhage, hepatitis, ileitis, jaundice, intestinal obstruction, liver tenderness, parotitis, periodontitis, proctitis, rectal disorder, salivary gland enlargement, increased salivation, soft stools, tongue discoloration.
Endocrine system - Rare: galactorrhoea, goiter, hyperthyroidism, hypothyroidism, thyroid nodule, thyroiditis.
Hemic and lymphatic system - Frequent: ecchymosis; Infrequent: anemia, leukocytosis, leukopenia, lymphadenopathy, thrombocythemia; Rare: basophilia, bleeding time increased, cyanosis, eosinophilia, lymphocytosis, multiple myeloma, purpura, thrombocytopenia.
Metabolic and nutritional - Frequent: edema, weight gain; Infrequent: alkaline phosphatase increased, dehydration, hypercholesteremia, hyperglycemia, hyperlipidemia, hypokalemia, SGOT (AST) increased, SGPT (ALT) increased, thirst; Rare: alcohol intolerance, bilirubinemia, BUN increased, creatinine increased, diabetes mellitus, glycosuria, gout, healing abnormal, hemochromatosis, hypercalcinuria, hyperkalemia, hyperphosphatemia, hyperuricemia, hypocholesteremia, hypoglycemia, hyponatremia, hypophosphatemia, hypoproteinemia, uremia.
Musculoskeletal system - Infrequent: arthritis, arthrosis, bone spurs, bursitis, leg cramps, myasthenia, tenosynovitis; Rare: bone pain, pathological fracture, muscle cramp, muscle spasms, musculoskeletal stiffness, myopathy, osteoporosis, osteosclerosis, plantar fasciitis, rheumatoid arthritis, tendon rupture.
Nervous system - Frequent: amnesia, confusion, depersonalization, hypesthesia, thinking abnormal, trismus, vertigo; Infrequent: akathisia, apathy, ataxia, circumoral paresthesia, CNS stimulation, emotional lability, euphoria, hallucinations, hostility, hyperesthesia, hyperkinesia, hypotonia, incoordination, libido increased, manic reaction, myoclonus, neuralgia, neuropathy, psychosis, seizure, abnormal speech, stupor, suicidal ideation; Rare: abnormal/changed behavior, adjustment disorder, akinesia, alcohol abuse, aphasia, bradykinesia, buccoglossal syndrome, cerebrovascular accident, feeling drunk, loss of consciousness, delusions, dementia, dystonia, energy increased, facial paralysis, abnormal gait, Guillain-Barre Syndrome, homicidal ideation, hyperchlorhydria, hypokinesia, hysteria, impulse control difficulties, motion sickness, neuritis, nystagmus, paranoid reaction, paresis, psychotic depression, reflexes decreased, reflexes increased, torticollis.
Respiratory system - Frequent: cough increased, dyspnea; Infrequent: asthma, chest congestion, epistaxis, hyperventilation, laryngismus, laryngitis, pneumonia, voice alteration; Rare: atelectasis, hemoptysis, hypoventilation, hypoxia, larynx edema, pleurisy, pulmonary embolus, sleep apnea.
Skin and appendages - Frequent: pruritus; Infrequent: acne, alopecia, contact dermatitis, dry skin, eczema, maculopapular rash, psoriasis, urticaria; Rare: brittle nails, erythema nodosum, exfoliative dermatitis, lichenoid dermatitis, hair discoloration, skin discoloration, furunculosis, hirsutism, leukoderma, miliaria, petechial rash, pruritic rash, pustular rash, vesiculobullous rash, seborrhea, skin atrophy, skin hypertrophy, skin striae, sweating decreased.
Special senses - Frequent: abnormality of accommodation, mydriasis, taste perversion; Infrequent: conjunctivitis, diplopia, dry eyes, eye pain, otitis media, parosmia, photophobia, taste loss; Rare: blepharitis, cataract, chromatopsia, conjunctival edema, corneal lesion, deafness, exophthalmos, eye hemorrhage, glaucoma, retinal hemorrhage, subconjunctival hemorrhage, hyperacusis, keratitis, labyrinthitis, miosis, papilledema, decreased pupillary reflex, otitis externa, scleritis, uveitis, visual field defect.
Urogenital system - Frequent: albuminuria, urination impaired; Infrequent: amenorrhea,* cystitis, dysuria, hematuria, kidney calculus, kidney pain, leukorrhea,* menorrhagia,* metrorrhagia,* nocturia, breast pain, polyuria, pyuria, prostatic disorder (prostatitis, enlarged prostate, and prostate irritability,* urinary incontinence, urinary retention, urinary urgency, vaginal hemorrhage,* vaginitis*; Rare: abortion,* anuria, breast discharge, breast engorgement, balanitis,* breast enlargement, endometriosis,* female lactation,* fibrocystic breast, calcium crystalluria, cervicitis,* orchitis,* ovarian cyst,* bladder pain, prolonged erection,* gynecomastia (male),* hypomenorrhea,* kidney function abnormal, mastitis, menopause,* pyelonephritis, oliguria, salpingitis,* urolithiasis, uterine hemorrhage,* uterine spasm,* vaginal dryness.*
* Based on the number of men and women as appropriate.
Postmarketing Reports
Adverse Events
Voluntary reports of other adverse events temporally associated with the use of venlafaxine that have been received since market introduction and that may have no causal relationship with the use of venlafaxine include the following: agranulocytosis, anaphylaxis, angioedema, aplastic anemia, catatonia, congenital anomalies, impaired coordination and balance, CPK increased, deep vein thrombophlebitis, delirium, EKG abnormalities such as QT prolongation; cardiac arrhythmias including atrial fibrillation, supraventricular tachycardia, ventricular extrasystoles, and rare reports of ventricular fibrillation and ventricular tachycardia, including torsade de pointes; toxic epidermal necrolysis/Stevens-Johnson Syndrome, erythema multiforme, extrapyramidal symptoms (including dyskinesia and tardive dyskinesia), angle-closure glaucoma, hemorrhage (including eye and gastrointestinal bleeding), hepatic events (including GGT elevation; abnormalities of unspecified liver function tests; liver damage, necrosis, or failure; and fatty liver), interstitial lung disease, involuntary movements, LDH increased, neutropenia, night sweats, pancreatitis, pancytopenia, panic, prolactin increased, renal failure, rhabdomyolysis, shock-like electrical sensations or tinnitus (in some cases, subsequent to the discontinuation of venlafaxine or tapering of dose), and syndrome of inappropriate antidiuretic hormone secretion (usually in the elderly).
Drug Interactions
There have been reports of elevated clozapine levels that were temporally associated with adverse events, including seizures, following the addition of venlafaxine. There have been reports of increases in prothrombin time, partial thromboplastin time, or INR when venlafaxine was given to patients receiving warfarin therapy.
DRUG ABUSE AND DEPENDENCE
Controlled Substance Class
Effexor XR (venlafaxine hydrochloride) extended-release capsules is not a controlled substance.
Physical and Psychological Dependence
In vitro studies revealed that venlafaxine has virtually no affinity for opiate, benzodiazepine, phencyclidine (PCP), or N-methyl-D-aspartic acid (NMDA) receptors.
Venlafaxine was not found to have any significant CNS stimulant activity in rodents. In primate drug discrimination studies, venlafaxine showed no significant stimulant or depressant abuse liability.
Discontinuation effects have been reported in patients receiving venlafaxine (see DOSAGE AND ADMINISTRATION).
While venlafaxine has not been systematically studied in clinical trials for its potential for abuse, there was no indication of drug-seeking behavior in the clinical trials. However, it is not possible to predict on the basis of premarketing experience the extent to which a CNS active drug will be misused, diverted, and/or abused once marketed. Consequently, physicians should carefully evaluate patients for history of drug abuse and follow such patients closely, observing them for signs of misuse or abuse of venlafaxine (eg, development of tolerance, incrementation of dose, drug-seeking behavior).
OVERDOSAGE
Human Experience
Among the patients included in the premarketing evaluation of Effexor XR, there were 2 reports of acute overdosage with Effexor XR in major depressive disorder trials, either alone or in combination with other drugs. One patient took a combination of 6 g of Effexor XR and 2.5 mg of lorazepam. This patient was hospitalized, treated symptomatically, and recovered without any untoward effects. The other patient took 2.85 g of Effexor XR. This patient reported paresthesia of all four limbs but recovered without sequelae.
There were 2 reports of acute overdose with Effexor XR in GAD trials. One patient took a combination of 0.75 g of Effexor XR and 200 mg of paroxetine and 50 mg of zolpidem. This patient was described as being alert, able to communicate, and a little sleepy. This patient was hospitalized, treated with activated charcoal, and recovered without any untoward effects. The other patient took 1.2 g of Effexor XR. This patient recovered and no other specific problems were found. The patient had moderate dizziness, nausea, numb hands and feet, and hot-cold spells 5 days after the overdose. These symptoms resolved over the next week.
There were no reports of acute overdose with Effexor XR in Social Anxiety Disorder trials.
There were 2 reports of acute overdose with Effexor XR in panic disorder trials. One patient took 0.675 g of Effexor XR once, and the other patient took 0.45 g of Effexor XR for 2 days. No signs or symptoms were associated with either overdose, and no actions were taken to treat them.
Among the patients included in the premarketing evaluation with Effexor (immediate release), there were 14 reports of acute overdose with venlafaxine, either alone or in combination with other drugs and/or alcohol. The majority of the reports involved ingestion in which the total dose of venlafaxine taken was estimated to be no more than several-fold higher than the usual therapeutic dose. The 3 patients who took the highest doses were estimated to have ingested approximately 6.75 g, 2.75 g, and 2.5 g. The resultant peak plasma levels of venlafaxine for the latter 2 patients were 6.24 and 2.35 μg/mL, respectively, and the peak plasma levels of O-desmethylvenlafaxine were 3.37 and 1.30 μg/mL, respectively. Plasma venlafaxine levels were not obtained for the patient who ingested 6.75 g of venlafaxine. All 14 patients recovered without sequelae. Most patients reported no symptoms. Among the remaining patients, somnolence was the most commonly reported symptom. The patient who ingested 2.75 g of venlafaxine was observed to have 2 generalized convulsions and a prolongation of QTc to 500 msec, compared with 405 msec at baseline. Mild sinus tachycardia was reported in 2 of the other patients.
In postmarketing experience, overdose with venlafaxine has occurred predominantly in combination with alcohol and/or other drugs. The most commonly reported events in overdosage include tachycardia, changes in level of consciousness (ranging from somnolence to coma), mydriasis, seizures, and vomiting. Electrocardiogram changes (eg, prolongation of QT interval, bundle branch block, QRS prolongation), ventricular tachycardia, bradycardia, hypotension, rhabdomyolysis, vertigo, liver necrosis, serotonin syndrome, and death have been reported.
Published retrospective studies report that venlafaxine overdosage may be associated with an increased risk of fatal outcomes compared to that observed with SSRI antidepressant products, but lower than that for tricyclic antidepressants. Epidemiological studies have shown that venlafaxine-treated patients have a higher pre-existing burden of suicide risk factors than SSRI-treated patients. The extent to which the finding of an increased risk of fatal outcomes can be attributed to the toxicity of venlafaxine in overdosage as opposed to some characteristic(s) of venlafaxine-treated patients is not clear. Prescriptions for Effexor XR should be written for the smallest quantity of capsules consistent with good patient management, in order to reduce the risk of overdose.
Management of Overdosage
Treatment should consist of those general measures employed in the management of overdosage with any antidepressant.
Ensure an adequate airway, oxygenation, and ventilation. Monitor cardiac rhythm and vital signs. General supportive and symptomatic measures are also recommended. Induction of emesis is not recommended. Gastric lavage with a large bore orogastric tube with appropriate airway protection, if needed, may be indicated if performed soon after ingestion or in symptomatic patients.
Activated charcoal should be administered. Due to the large volume of distribution of this drug, forced diuresis, dialysis, hemoperfusion, and exchange transfusion are unlikely to be of benefit. No specific antidotes for venlafaxine are known.
In managing overdosage, consider the possibility of multiple drug involvement. The physician should consider contacting a poison control center for additional information on the treatment of any overdose. Telephone numbers for certified poison control centers are listed in the Physicians' Desk Reference® (PDR).
DOSAGE AND ADMINISTRATION
Effexor XR should be administered in a single dose with food either in the morning or in the evening at approximately the same time each day. Each capsule should be swallowed whole with fluid and not divided, crushed, chewed, or placed in water, or it may be administered by carefully opening the capsule and sprinkling the entire contents on a spoonful of applesauce. This drug/food mixture should be swallowed immediately without chewing and followed with a glass of water to ensure complete swallowing of the pellets.
Initial Treatment
Major Depressive Disorder
For most patients, the recommended starting dose for Effexor XR is 75 mg/day, administered in a single dose. In the clinical trials establishing the efficacy of Effexor XR in moderately depressed outpatients, the initial dose of venlafaxine was 75 mg/day. For some patients, it may be desirable to start at 37.5 mg/day for 4 to 7 days, to allow new patients to adjust to the medication before increasing to 75 mg/day. While the relationship between dose and antidepressant response for Effexor XR has not been adequately explored, patients not responding to the initial 75 mg/day dose may benefit from dose increases to a maximum of approximately 225 mg/day. Dose increases should be in increments of up to 75 mg/day, as needed, and should be made at intervals of not less than 4 days, since steady state plasma levels of venlafaxine and its major metabolites are achieved in most patients by day 4. In the clinical trials establishing efficacy, upward titration was permitted at intervals of 2 weeks or more; the average doses were about 140 to 180 mg/day (see Clinical Trials under CLINICAL PHARMACOLOGY).
It should be noted that, while the maximum recommended dose for moderately depressed outpatients is also 225 mg/day for Effexor (immediate release), more severely depressed inpatients in one study of the development program for that product responded to a mean dose of 350 mg/day (range of 150 to 375 mg/day). Whether or not higher doses of Effexor XR are needed for more severely depressed patients is unknown; however, the experience with Effexor XR doses higher than 225 mg/day is very limited. (See PRECAUTIONS-General-Use in Patients with Concomitant Illness.)
Generalized Anxiety Disorder
For most patients, the recommended starting dose for Effexor XR is 75 mg/day, administered in a single dose. In clinical trials establishing the efficacy of Effexor XR in outpatients with Generalized Anxiety Disorder (GAD), the initial dose of venlafaxine was 75 mg/day. For some patients, it may be desirable to start at 37.5 mg/day for 4 to 7 days, to allow new patients to adjust to the medication before increasing to 75 mg/day. Although a dose-response relationship for effectiveness in GAD was not clearly established in fixed-dose studies, certain patients not responding to the initial 75 mg/day dose may benefit from dose increases to a maximum of approximately 225 mg/day. Dose increases should be in increments of up to 75 mg/day, as needed, and should be made at intervals of not less than 4 days. (See the Use in Patients with Concomitant Illness section of PRECAUTIONS.)
Social Anxiety Disorder (Social Phobia)
The recommended dose is 75 mg/day, administered in a single dose. There was no evidence that higher doses confer any additional benefit. (See the Use in Patients with Concomitant Illness section of PRECAUTIONS.)
Panic Disorder
It is recommended that initial single doses of 37.5 mg/day of Effexor XR be used for 7 days. In clinical trials establishing the efficacy of Effexor XR in outpatients with panic disorder, initial doses of 37.5 mg/day for 7 days were followed by doses of 75 mg/day and subsequent weekly dose increases of 75 mg/day to a maximum dose of 225 mg/day. Although a dose-response relationship for effectiveness in patients with panic disorder was not clearly established in fixed-dose studies, certain patients not responding to 75 mg/day may benefit from dose increases to a maximum of approximately 225 mg/day. Dose increases should be in increments of up to 75 mg/day, as needed, and should be made at intervals of not less than 7 days. (See the Use in Patients with Concomitant Illness section of PRECAUTIONS.)
Switching Patients from Effexor Tablets
Depressed patients who are currently being treated at a therapeutic dose with Effexor (immediate release) may be switched to Effexor XR at the nearest equivalent dose (mg/day), eg, 37.5 mg venlafaxine two-times-a-day to 75 mg Effexor XR once daily. However, individual dosage adjustments may be necessary.
Special Populations
Treatment of Pregnant Women During the Third Trimester
Neonates exposed to Effexor XR, other SNRIs, or SSRIs, late in the third trimester have developed complications requiring prolonged hospitalization, respiratory support, and tube feeding (see PRECAUTIONS). When treating pregnant women with Effexor XR during the third trimester, the physician should carefully consider the potential risks and benefits of treatment. The physician may consider tapering Effexor XR in the third trimester.
Patients with Hepatic Impairment
Given the decrease in clearance and increase in elimination half-life for both venlafaxine and ODV that is observed in patients with hepatic cirrhosis and mild and moderate hepatic impairment compared with normal subjects (see CLINICAL PHARMACOLOGY), it is recommended that the total daily dose be reduced by 50% in patients with mild to moderate hepatic impairment. Since there was much individual variability in clearance between subjects with cirrhosis, it may be necessary to reduce the dose even more than 50%, and individualization of dosing may be desirable in some patients.
Patients with Renal Impairment
Given the decrease in clearance for venlafaxine and the increase in elimination half-life for both venlafaxine and ODV that is observed in patients with renal impairment (GFR = 10 to 70 mL/min) compared with normal subjects (see CLINICAL PHARMACOLOGY), it is recommended that the total daily dose be reduced by 25% to 50%. In patients undergoing hemodialysis, it is recommended that the total daily dose be reduced by 50%. Because there was much individual variability in clearance between patients with renal impairment, individualization of dosage may be desirable in some patients.
Elderly Patients
No dose adjustment is recommended for elderly patients solely on the basis of age. As with any drug for the treatment of major depressive disorder, Generalized Anxiety Disorder, Social Anxiety Disorder, or panic disorder, however, caution should be exercised in treating the elderly. When individualizing the dosage, extra care should be taken when increasing the dose.
Maintenance Treatment
There is no body of evidence available from controlled trials to indicate how long patients with major depressive disorder, Generalized Anxiety Disorder, Social Anxiety Disorder, or panic disorder, should be treated with Effexor XR.
It is generally agreed that acute episodes of major depressive disorder require several months or longer of sustained pharmacological therapy beyond response to the acute episode. In one study, in which patients responding during 8 weeks of acute treatment with Effexor XR were assigned randomly to placebo or to the same dose of Effexor XR (75, 150, or 225 mg/day, qAM) during 26 weeks of maintenance treatment as they had received during the acute stabilization phase, longer-term efficacy was demonstrated. A second longer-term study has demonstrated the efficacy of Effexor in maintaining a response in patients with recurrent major depressive disorder who had responded and continued to be improved during an initial 26 weeks of treatment and were then randomly assigned to placebo or Effexor for periods of up to 52 weeks on the same dose (100 to 200 mg/day, on a b.i.d. schedule) (see Clinical Trials under CLINICAL PHARMACOLOGY). Based on these limited data, it is not known whether or not the dose of Effexor/Effexor XR needed for maintenance treatment is identical to the dose needed to achieve an initial response. Patients should be periodically reassessed to determine the need for maintenance treatment and the appropriate dose for such treatment.
In patients with Generalized Anxiety Disorder, Effexor XR has been shown to be effective in 6-month clinical trials. The need for continuing medication in patients with GAD who improve with Effexor XR treatment should be periodically reassessed.
In patients with Social Anxiety Disorder, Effexor XR has been shown to be effective in a 6-month clinical trial. The need for continuing medication in patients with Social Anxiety Disorder who improve with Effexor XR treatment should be periodically reassessed.
In a study of panic disorder in which patients responding during 12 weeks of acute treatment with Effexor XR were assigned randomly to placebo or to the same dose of Effexor XR (75, 150, or 225 mg/day), patients continuing Effexor XR experienced a significantly longer time to relapse than patients randomized to placebo. The need for continuing medication in patients with panic disorder who improve with Effexor XR treatment should be periodically reassessed.
Discontinuing Effexor XR
Symptoms associated with discontinuation of Effexor XR, other SNRIs, and SSRIs, have been reported (see PRECAUTIONS). Patients should be monitored for these symptoms when discontinuing treatment. A gradual reduction in the dose rather than abrupt cessation is recommended whenever possible. If intolerable symptoms occur following a decrease in the dose or upon discontinuation of treatment, then resuming the previously prescribed dose may be considered. Subsequently, the physician may continue decreasing the dose but at a more gradual rate. In clinical trials with Effexor XR, tapering was achieved by reducing the daily dose by 75 mg at 1 week intervals. Individualization of tapering may be necessary.
Switching Patients To or From a Monoamine Oxidase Inhibitor
At least 14 days should elapse between discontinuation of an MAOI and initiation of therapy with Effexor XR. In addition, at least 7 days should be allowed after stopping Effexor XR before starting an MAOI (see CONTRAINDICATIONS and WARNINGS).
HOW SUPPLIED
Effexor XR® (venlafaxine hydrochloride) extended-release capsules are available as follows:
37.5 mg, grey cap/peach body with W and “Effexor XR” on the cap and “37.5” on the body.
- NDC 0008-0837-20, bottle of 15 capsules in unit of use package
- NDC 0008-0837-21, bottle of 30 capsules in unit of use package.
- NDC 0008-0837-22, bottle of 90 capsules in unit of use package.
- NDC 0008-0837-03, carton of 10 Redipak® blister strips of 10 capsules each.
75 mg, peach cap and body with W and “Effexor XR” on the cap and “75” on the body.
- NDC 0008-0833-20, bottle of 15 capsules in unit of use package.
- NDC 0008-0833-21, bottle of 30 capsules in unit of use package.
- NDC 0008-0833-22, bottle of 90 capsules in unit of use package.
- NDC 0008-0833-03, carton of 10 Redipak® blister strips of 10 capsules each.
150 mg, dark orange cap and body with W and “Effexor XR” on the cap and “150” on the body.
- NDC 0008-0836-20, bottle of 15 capsules in unit of use package.
- NDC 0008-0836-21, bottle of 30 capsules in unit of use package.
- NDC 0008-0836-22, bottle of 90 capsules in unit of use package.
- NDC 0008-0836-03, carton of 10 Redipak® blister strips of 10 capsules each.
Store at controlled room temperature, 20° to 25°C (68° to 77°F).
The unit of use package is intended to be dispensed as a unit.
The appearance of these capsules is a trademark of Wyeth Pharmaceuticals.
U.S. Patent Nos. 5,916,923; 6,274,171; 6,310,101; 6,403,120; 6,419,958; and 6,444,708.
MEDICATION GUIDE
Antidepressant Medicines, Depression and Other Serious Mental Illnesses, and Suicidal Thoughts or Actions
Read the Medication Guide that comes with your or your family member's antidepressant medicine. This Medication Guide is only about the risk of suicidal thoughts and actions with antidepressant medicines.
Talk to your, or your family member's, healthcare provider about:
- all risks and benefits of treatment with antidepressant medicines
- all treatment choices for depression or other serious mental illness
What is the most important information I should know about antidepressant medicines, depression and other serious mental illnesses, and suicidal thoughts or actions?
1. Antidepressant medicines may increase suicidal thoughts or actions in some children, teenagers, and young adults within the first few months of treatment.
2. Depression and other serious mental illnesses are the most important causes of suicidal thoughts and actions. Some people may have a particularly high risk of having suicidal thoughts or actions. These include people who have (or have a family history of) bipolar illness (also called manic-depressive illness) or suicidal thoughts or actions.
3. How can I watch for and try to prevent suicidal thoughts and actions in myself or a family member?
- Pay close attention to any changes, especially sudden changes, in mood, behaviors, thoughts, or feelings. This is very important when an antidepressant medicine is started or when the dose is changed.
- Call the healthcare provider right away to report new or sudden changes in mood, behavior, thoughts, or feelings.
- Keep all follow-up visits with the healthcare provider as scheduled. Call the healthcare provider between visits as needed, especially if you have concerns about symptoms.
Call a healthcare provider right away if you or your family member has any of the following symptoms, especially if they are new, worse, or worry you:
|
|
|
|
|
|
|
|
|
|
|
|
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
What else do I need to know about antidepressant medicines?
- Never stop an antidepressant medicine without first talking to a healthcare provider. Stopping an antidepressant medicine suddenly can cause other symptoms.
- Antidepressants are medicines used to treat depression and other illnesses. It is important to discuss all the risks of treating depression and also the risks of not treating it. Patients and their families or other caregivers should discuss all treatment choices with the healthcare provider, not just the use of antidepressants.
- Antidepressant medicines have other side effects. Talk to the healthcare provider about the side effects of the medicine prescribed for you or your family member.
- Antidepressant medicines can interact with other medicines. Know all of the medicines that you or your family member takes. Keep a list of all medicines to show the healthcare provider. Do not start new medicines without first checking with your healthcare provider.
- Not all antidepressant medicines prescribed for children are FDA approved for use in children. Talk to your child's healthcare provider for more information.
This Medication Guide has been approved by the U.S. Food and Drug Administration for all antidepressants.
Official site